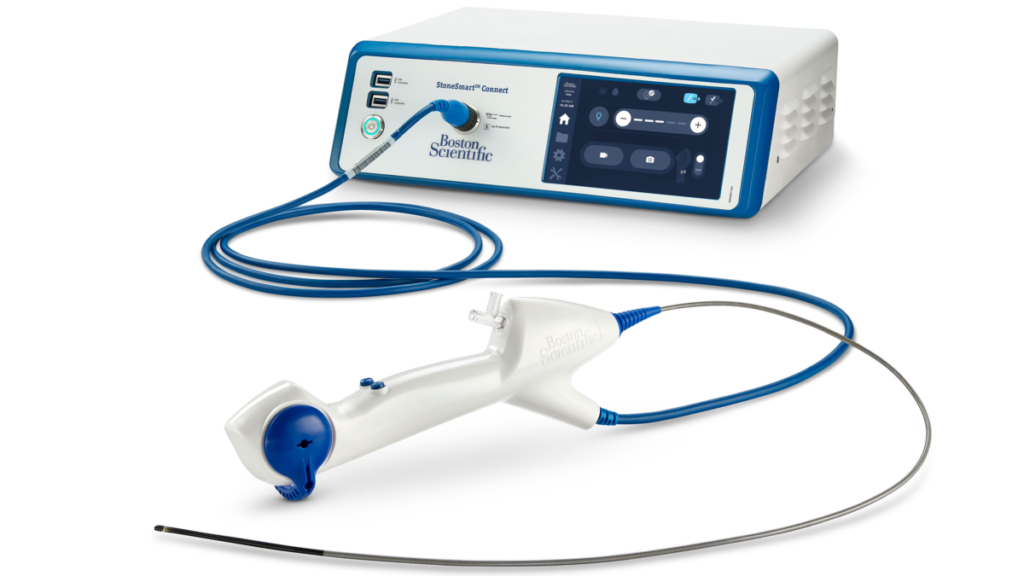
Boston Scientific’s new LithoVue Elite single-use digital flexible ureteroscope system recently received 510(k) clearance from the US Food and Drug Administration (FDA). This ureteroscope is the first of its kind, as it can observe intrarenal pressure (IRP) in real-time during a ureteroscopy procedure. The LithoVue Elite System comes with the StoneSmart Connect Console.
The LithoVue Elite System was developed with physician feedback to improve patient care and stop infection risk from the unsuccessful reprocessing of reusable scopes.
Ureteroscopy is a common procedure done by urologists to help examine kidney stones, tumors in the renal system or inspect an infection. This process can be painful as it can lead to bleeding or swelling.
During a ureteroscopy, fluid irrigation is used to keep a clear visual field, but this can cause elevated IRP. There could be several post-operative complications from high IRP, including sepsis, renal damage, pyelovenous backflow and systemic inflammatory response syndrome (SIRS). To mitigate these problems, the LithoVue Elite ureteroscope tip has a built-in pressure sensor to allow urologists to make real-time pressure-related clinical decisions.
“Multiple studies have shown the importance of understanding IRP during ureteroscopy procedures to mitigate potential complications, but until now, urologists have lacked an easy way to measure pressure in real-time,” said Ben Chew, MD, MSc, associate professor, University of British Columbia, in the company’s press release.
“This device represents an important and needed advancement in single-use ureteroscopes, which could help improve patient care and potentially lead to a clearer understanding of the impact of elevated intrarenal pressure on patient outcomes,” added Dr. Chew.
XTALKS WEBINAR: EU MDR: Compliance Requirements From the Pharmacovigilance and Vigilance Perspective
Live and On-Demand: Tuesday, March 14, 2023, at 10am EDT (3pm CET/EU-Central)
Register for this free webinar to learn about the nuances and requirements of the latest European Medical Device Regulation (EU MDR). Attendees will learn how drug and device vigilance requirements compare, and the reporting requirements for both clinical investigations and postmarketing.
LithoVue Elite: A Unique and Safe Ureteroscope
The LithoVue Elite system has distinct features to improve ureteroscopy procedures, as it:
- gives bright, clear and sharp images with accurate, bold colors and fast image processing for immediate visualization,
- has a digital CMOS imager to give a clear image with a depth of view of 2 mm to 50 mm,
- brings a wide view and 270° deflection in both directions,
- has an integrated LED light source,
- has an all-in-one touchscreen to monitor and process the images.
In addition to monitoring IRP, the newest LithoVuew Elite edition has upgraded image quality, updated control features and streamlined integration with existing endoscopic monitors and operating room visualization towers.
“We’re at a critical moment in kidney stone care where patient demand is outpacing the number of urologists needed to address it,” said Meghan Scanlon, senior vice president and president, Urology, Boston Scientific, in the press release.
“We designed the next-generation LithoVue Elite System to bring a greater level of precision and functionality in the way urologists diagnose and treat kidney stones, an increasingly common condition among adults. We are committed to developing clinical solutions, like the LithoVue Elite System, that are intended to make procedures more efficient, empower surgical decisions, optimize patient care and ultimately improve the treatment of kidney stone disease globally,” added Scanlon.











Join or login to leave a comment
JOIN LOGIN