At the Digital International Liver Congress 2020, which was held virtually from August 27 – 29, 2020, Viking Therapeutics, Inc. presented promising results from a 12-week Phase II study of its small molecule compound VK2809 in patients with non-alcoholic fatty liver disease (NAFLD) and elevated low-density lipoprotein cholesterol (LDL-C). The release of the data was presented as the flagship event for the annual meeting of the European Association for the Study of the Liver (EASL).
Viking Therapeutics is a clinical-stage biopharmaceutical company that develops novel therapies for metabolic and endocrine disorders.
Viking Therapeutics’ oral presentation at the meeting included newly reported data which showed that patients treated with VK2809 experienced significant and durable reductions in liver fat content that were maintained at Week 16, four weeks after administration of the last dose in the 12-week study. The study also demonstrated that liver fat content was significantly decreased in patients that received VK2809, regardless of baseline characteristics and common risk factors for non-alcoholic steatohepatitis (NASH) such as Hispanic ethnicity, hypertension or a body mass index (BMI) greater than 30.
It is estimated that NAFLD affects approximately 25 percent of the global population, with an estimated one billion people currently afflicted with the disease. Its incidence can be as high as 90 percent in those that are obese. It is now seen as an epidemic within chronic liver disease, particularly as its prevalence has been increasing and paralleling that of obesity, making it a major health concern worldwide.
Related: Six of the Latest NASH/NAFLD Webinars
VK2809 is a novel, oral small molecule thyroid hormone receptor beta agonist that has selectivity for liver tissue. It was developed by Viking Therapeutics for the treatment of lipid and metabolic disorders.
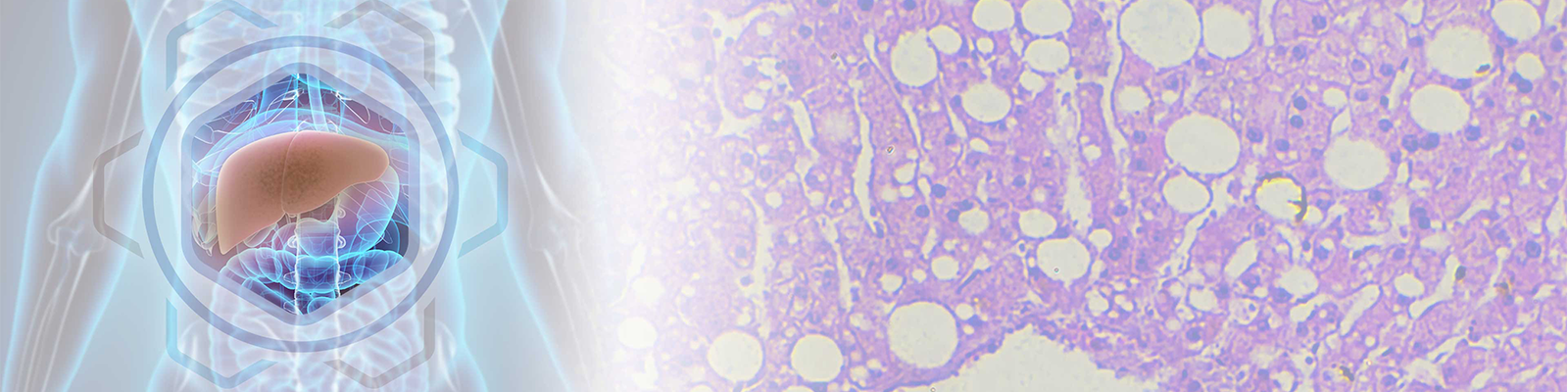
According to the American College of Gastroenterology (ACG), NAFLD refers to a group of disorders that involves accumulation of excess fat in the liver of people who drink little or no alcohol. The condition; known as ‘fatty liver;’ is the most common and non-serious form of NAFLD, and occurs when fat accumulates in liver cells. Although the presence of fat in the liver is not normal, it likely does not damage the organ on its own.
On the other hand, a small number of people with NAFLD may have a more serious condition called non-alcoholic steatohepatitis (NASH). In NASH, fat accumulation is associated with liver cell inflammation and different degrees of scarring. NASH may result in severe liver scarring and cirrhosis.
NAFLD is part of the metabolic syndrome that is characterized by diabetes, or pre-diabetes (insulin resistance), which includes being overweight or obese, elevated blood lipids such as cholesterol and triglycerides, as well as high blood pressure. The causes of NASH remain largely unknown but factors such as oxidative stress, excess production of inflammatory cytokines, liver necrosis, inflammation of adipose tissue and the gut microflora may contribute to its development.
VK2809 and the VOYAGE Trial
In 2019, Viking Therapeutics began a multicenter Phase IIb randomized, double-blind, placebo-controlled study called the VOYAGE trial to evaluate the safety, efficacy and tolerability of VK2809 in patients with biopsy-confirmed NASH and fibrosis. The trial’s primary endpoint assessed the effect of VK2809 treatment on LDL-C after 12 weeks compared to placebo, while the secondary endpoint evaluated changes in liver fat content by magnetic resonance imaging, proton density fat fraction (MRI-PDFF).
The study had a target enrollment of approximately 340 patients across five treatment arms: 1.0 mg daily; 2.5 mg daily; 5.0 mg every other day; 10.0 mg every other day; and placebo.
Results from the trial showed that patients that were administered VK2809 demonstrated a statistically significant 45.4 percent median reduction in liver fat content at Week 16 compared to an 18.7 percent reduction for placebo.
These treatment effects were robust and durable as they were maintained beyond the last treatment dose. Specifically, at Week 16 — four weeks after administration of the last dose (at Week 12) — 70.4 percent of all patients that had received VK2809 demonstrated ≥ 30 percent relative liver fat reduction from baseline compared to 22.2 percent for placebo-treated patients. Notably, 100 percent of the patients that received the daily 5.0 mg dose of VK2809 were still considered responders at Week 16.
“We are pleased with the durability of therapeutic response achieved with VK2809 in this study. Not only did patients receiving VK2809 maintain significant liver fat reductions many weeks after their final study dose, but more than 70 percent of all VK2809-treated patients still qualified as responders four weeks post-treatment,” said Brian Lian, PhD, chief executive officer of Viking in a press statement from the company.
Previous data from the study showed that VK2809 successfully achieved both its primary and secondary efficacy endpoints, with median relative reductions in liver fat ranging from approximately 54 to 60 percent, and response rates of up to 100 percent, which is unprecedented efficacy demonstrated by an oral agent. VK2809 was shown to be safe and well tolerated at all doses evaluated and no serious adverse events were reported.
“These latest results offer further support for what we believe is a best-in-class therapeutic profile for VK2809 and we look forward to completing our ongoing Phase IIb VOYAGE study in patients with biopsy-confirmed NASH,” said Lian.
The results from the study provide a strong basis for the further development of VK2809 for the treatment of NASH and related inflammation-associated liver conditions.




Join or login to leave a comment
JOIN LOGIN