- How can stronger relationships be built between sponsors and CROs?
- How can the sharing of trial data improve trial outcomes?
- What are the best ways to measure performance and conduct between the sponsor and CRO?
These are top challenges for sponsors and CROs today. In light of the ICH E6(R2) guidelines, sponsors and CROs are reviewing and assessing how well they work together.
This webinar will present a detailed look at the interactions and performance of sponsor-CRO driven studies. Paragon, a leading consulting firm that helps health and life sciences companies become high-performing, compliant and digitally connected, will discuss their hands-on perspective of these interactions and provide best practice advice on how to address the three key ways that the new guidelines impact study conduct. Comprehend Systems will then offer a short case study of how one sponsor tackled the ICH E6(R2) guidelines while measurably improving their relationships with multiple CROs.
The webinar will focus on issues such as:
- Steps for assessing how aligned your organization is with R2 expectations for study oversight
- Benefits of meeting R2 requirements with a risk-based oversight approach
- How automation helps reduce the time and effort to improve performance, reduce risk and see across data
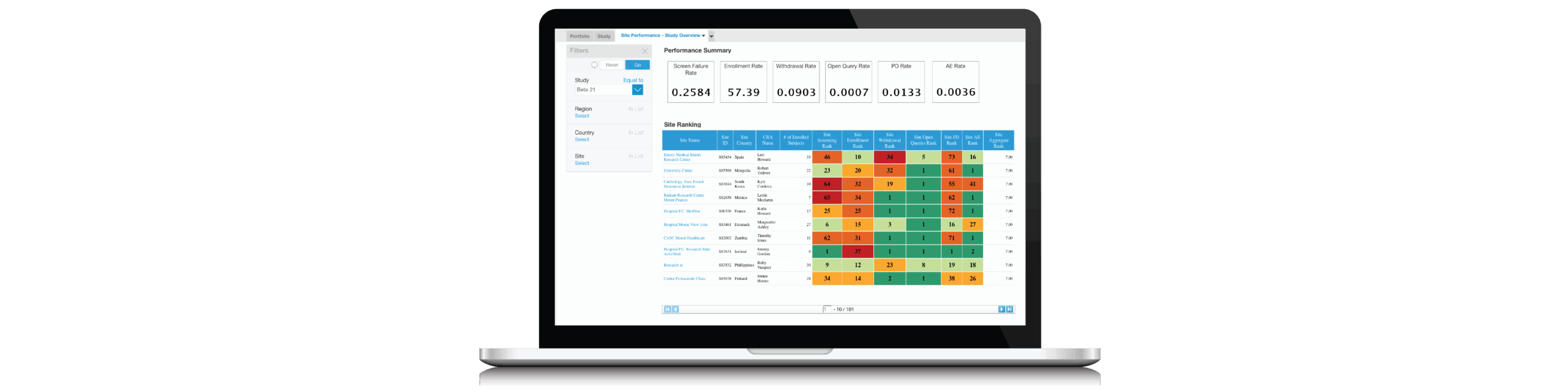
Karen McCarthy Schau of Paragon will examine the Best Practices of sponsor-CRO relationships and performance. Julie Peacock of Comprehend will follow with a case study and short demonstration of automation used by the sponsor and CRO to:
- Monitor Quality Agreement metrics
- Proactively identify and mitigate risk
- Track portfolio and site performance
- Empower teams through collaboration and workflow
Speakers

Julie Peacock, Client Services, Comprehend
Closely aligned with customers and the sales organization, Julie focuses on enabling prospects and customers on Comprehend’s Clinical Intelligence solutions. She manages go to market strategy, sales enablement, and product marketing.
Prior to Comprehend, Julie spent 18 years at Oracle Corporation in the enterprise application space where she managed strategy, field enablement and launch activities for a series of BtoB solutions. Julie holds a bachelor’s degree in Marketing from Auburn University.

Karen McCarthy Schau, Associate Director, R&D Consulting Group, Paragon
Karen McCarthy Schau is an Associate Director in Paragon’s R&D Consulting group and joined Paragon in 2014. Karen has 19 years of diversified experience across functional areas with an emphasis on Clinical Systems, and more than 10 years specializing in process reengineering and system implementation for a large global CRO. Karen currently focuses on helping companies deploy solutions to streamline processes, improve productivity, and operational efficiency, and provides leadership and support for system implementations. She is a member of the MCC Metrics Consortium.
Who Should Attend?
CROs, Clinical Operations and Data Management Professionals
- Clinical Trial/Clinical Study Management
- Clinical Data/Informatics/IT
- Clinical Outsourcing
Clinical Research, Technology and Business Professionals
- Biometrics/Biostatistics
- Business Technology/Applications/Solutions
- Business Analyst
- CTO
- Project Management
Xtalks Partners
Comprehend
Comprehend offers a suite of Clinical Intelligence applications that enables ClinOps Execs, Data Managers and Medical Monitors to significantly improve the speed, safety and quality of a portfolio of clinical trials. Across studies, sites, systems and CROs, Comprehend’s Clinical Intelligence Suite is particularly effective for centralized monitoring, risk monitoring, CRO oversight and collaboration, and medical monitoring initiatives. Comprehend gives life sciences companies a new source of competitiveness and the confidence to deliver high quality trial submissions at a new speed. Comprehend: the speed to quality results. Learn more at www.comprehend.com
Paragon
Paragon is a consulting firm, now part of CGI, that helps health and life sciences companies become high-performing, compliant, and digitally connected. Paragon powers business transformation and delivers better business outcomes by providing valuable consulting services as a trusted partner to our clients. We do this by building long-term client relationships based on our domain expertise, creative ideas, pragmatic consulting services, and quality delivery of solutions.
You Must Login To Register for this Free Webinar
Already have an account? LOGIN HERE. If you don’t have an account you need to create a free account.
Create Account