World Hearing Day 2026 is observed on March 3. Led by the WHO, the day is meant to raise awareness about hearing loss, ear care and access to hearing services worldwide.
From consumer devices like Apple AirPods Pro with built-in hearing tests to prescription hearing aids such as Oticon’s latest systems that support Google Fast Pair on Android devices, hearing support technologies are becoming more accessible and more capable.
However, there still remains a gap. With around 90 million children and adolescents affected by hearing loss worldwide, many of whom remain undiagnosed, the WHO implores 2026 with the theme, “From communities to classrooms: hearing care for all children,” which focuses on preventing avoidable childhood hearing loss and improving early identification and care, particularly for children.
The Scope of Hearing Loss in Children
Globally, the WHO estimates that 430 million people currently live with disabling hearing loss. By 2050, that number is expected to jump to 2.5 billion people having some degree of hearing loss, with more than 700 million requiring rehabilitation.
Population aging, increased noise exposure and gaps in access to care are contributing factors.
For children, delayed diagnosis can affect development. In the US, about 1 in 500 infants is born deaf or hard of hearing, making hearing loss one of the most common congenital conditions. National Health and Nutrition Examination Survey (NHANES III) data suggested that adolescents aged 12 to 19 have some degree of hearing loss.
Common causes of childhood hearing loss include otitis media (middle-ear infection), vaccine-preventable infections, excessive noise exposure and untreated ear conditions such as impacted earwax. When hearing loss is not addressed early, it can affect learning and social development well into adulthood.
Device and Surgical Advances Expanding Pediatric Access
Recent regulatory and clinical developments are supporting earlier intervention in pediatric hearing care.
In December 2025, the FDA approved an expanded indication for MED-EL cochlear implant systems for children as young as seven months with bilateral sensorineural hearing loss. Clinical data showed the implants were safe and effective in infants and toddlers, with no increase in major complications in children implanted before 12 months of age.
In January 2026, the FDA cleared iotaMotion’s iotaSOFT robotic-assisted cochlear implant insertion system for use in children aged four years and older, expanding its pediatric indication. The system enables slow, controlled insertion of the electrode array into the inner ear (cochlea), a step intended to help preserve delicate structures involved in hearing.
Very recently, NYU Langone Health reported the case of the first US pediatric patient to receive the FDA-approved Nucleus Nexa cochlear implant system from Cochlear Limited, implanted at 18 months of age following a diagnosis of severe bilateral sensorineural hearing loss. The system features upgradable firmware, meaning software updates can be applied without replacing the device.
Innovation in Implant Design and Hearing Aids
Manufacturers are introducing design changes to improve how hearing devices are used over time.
In late 2025, Smart Sound Ltd. published a US patent describing a hidden, magnet-free cochlear implant system. The design places sound processing in an in-canal transmitter and uses a conventional internal implant. The system does not rely on an external on-the-head unit and is designed to transmit power and data to the implant.
Oticon has also expanded its hearing aid portfolio with smaller device designs. In October 2025, Oticon announced the Oticon Intent miniBTE R, a rechargeable hearing aid that uses sensor-based sound processing informed by movement and the surrounding sound environment.
Beyond Devices: Gene Therapy Enters the Hearing Pipeline
Hearing health innovation is also extending into biotechnology. In January 2026, Sensorion announced a €60 million (close to $71 million) financing round, including a strategic investment from Sanofi, to advance its genetic medicine pipeline for hearing loss. Key programs include SENS-601 (GJB2-GT), which targets hearing loss associated with GJB2 mutations. These mutations account for roughly 50% of autosomal recessive non-syndromic congenital hearing loss and are also linked to early-onset forms of severe hearing loss in adults.
Sensorion is also advancing SENS-501, which entered clinical development in 2024. While investigational, these programs show how genetic approaches are being explored to combat hearing loss.
If you want your company to be featured on Xtalks.com, please email [email protected].

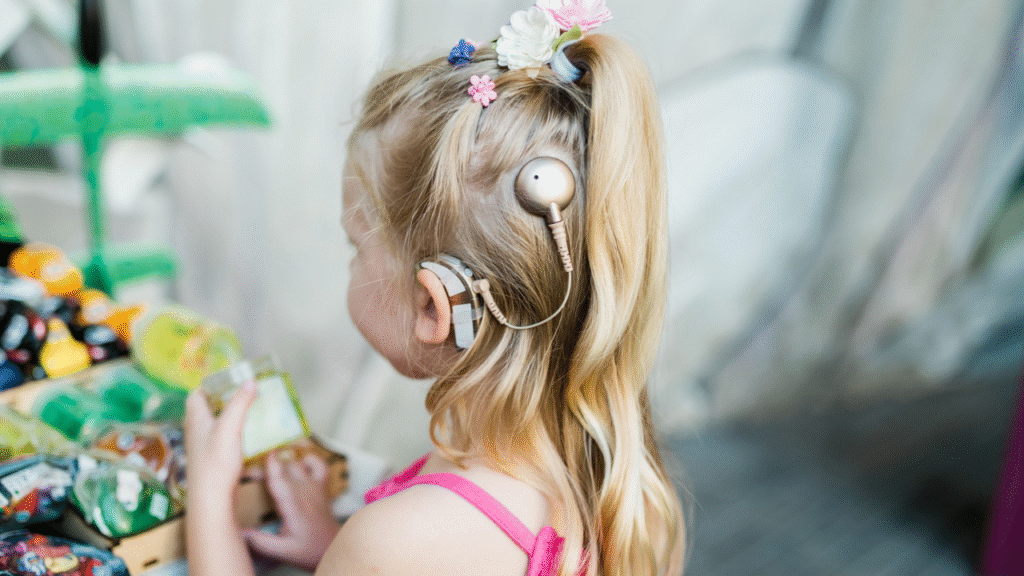











Join or login to leave a comment
JOIN LOGIN