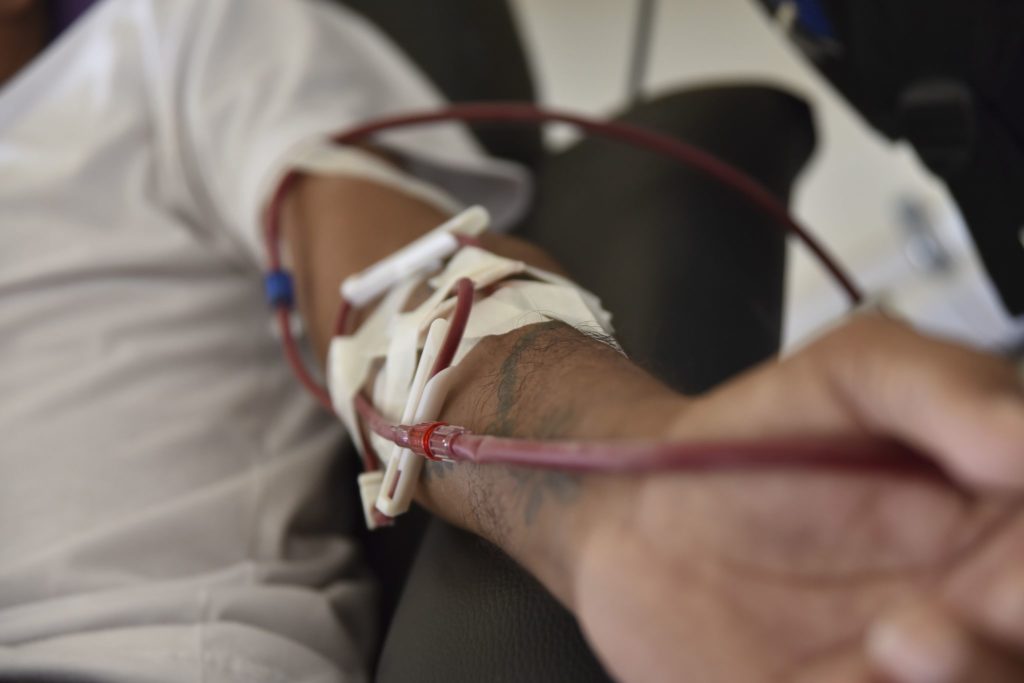
A decellularized blood vessel graft developed by researchers at the University of Minnesota could improve the lives of tens of thousands of kidney dialysis patients in the US. That’s because the graft is the first nonsynthetic blood vessel on which a patient’s cells can grow after transplantation.
In a recent preclinical study in nonhuman primates, the blood vessel graft performed well in terms of tolerability and functionality. According to the researchers – who published the results of this study in the journal, Science Translational Medicine – say the lab-grown vessel could act as an “off the shelf” graft for patients undergoing kidney dialysis.
Most of the over 400,000 patients who are treated using hemodialysis in the US rely on a surgically-created arteriovenous fistula in the arm to provide blood vessel access. But for as many as half of these patients, an artificial arteriovenous graft is required to overcome complications with the preferred method.
Since artificial grafts can increase the risk of clotting and infection, researchers have been working on developing living grafts made from biological tissue. However, these grafts must be made using a patient’s own cells to avoid possible rejection, presenting a serious barrier to large-scale commercialization of these living grafts.
The University of Minnesota researchers sought to overcome this hurdle by growing grafts using a blood clotting protein known as cow fibrin, and human skin cells which were removed prior to transplantation. By allowing these components to incubate in a bioreactor for a period of seven weeks, they were able to generate a graft containing all the necessary biomaterials for a working blood vessel, while still being able to remove the cells at the end.
“We harnessed the body’s normal wound-healing system in this process by starting with skin cells in a fibrin gel, which is nature’s starting point for healing,” said University of Minnesota Department of Biomedical Engineering Professor Robert Tranquillo. “Washing away the cells in the final step reduces the chance of rejection. This also means the vessels can be stored and implanted when they are needed because they are no longer a living material. In the future, thousands of the lab-grown vessels could be made from a small skin biopsy from one donor and then stored on the shelf for when they are needed by patients.”
But generating the blood vessel grafts was only half the battle: Tranquillo and his colleagues implanted the grafts into adult baboons to assess how well they would function in a recipient. They found that the grafts were populated with the baboons’ cells within 6 months of them being implanted, and they were strong enough to withstand 30 times the average human blood pressure.
In addition to be well-tolerated by the subjects, the grafts displayed no evidence of calcification – a common problem with synthetic grafts. Importantly, the grafts were able to self-heal puncture wounds similar to those required for regular hemodialysis.
“This preclinical trial was extremely important to us,” Tranquillo said. “In previous studies, we implanted vessels in sheep, but we needed to test them in a more human-like model before risking human lives because the success of our material depends on the ability of the recipient to recellularize it into a living tissue without immune response, which might have failed in a human even though it succeeded in a sheep.”
These blood vessel grafts could also be tailored for different applications, such as coronary and peripheral replacement vessels. Tranquillo and his colleagues plan to conduct a clinical trial in pediatric heart patients using the same techniques to grow the grafts.





Join or login to leave a comment
JOIN LOGIN