The year 2020 is certainly one that will go down in the books, thanks — or rather, no thanks — to the COVID-19 pandemic. The pandemic thrust the life science and healthcare sectors onto center stage, and they rose to the occasion against the most unprecedented health challenge in recent times.
For some, widespread disease outbreaks may have been seen as a thing of the past, or part of the storyline of a Hollywood movie. However, SARS-CoV-2 quickly changed that, taking the world by storm with lockdowns, mask mandates, building of temporary hospitals and hard looks at pandemic preparedness and how we must better safeguard vulnerable populations like the elderly and disadvantaged, among many, many other things.
The most positive thing to be gleaned from the pandemic is how it allowed for the power and pursuit of science to shine. While there may have been some initial distrust with respect to confusing messaging on the part of health and government officials, trust in science, on the other hand, has largely continued to prevail and lead the way forward.
From isolating SARS-CoV-2 in early January to sequencing its genome shortly thereafter and having a prototype vaccine against it within days, scientific process and progress have held steadfast throughout the pandemic.
Infectious disease expert, epidemiologist and director of the Yale Institute for Global Health at Yale University, Saad B. Omer, MBBS, MPH, PhD, told Xtalks it is important to “invest in platform technologies before pandemics or interpandemics happen.”
He explains, “For example, investment in mRNA vaccines and viral vectors came well before January, many years before it. Focusing on biologics, vaccine development and policy platforms in anticipation of an eventual outbreak is why we’re where we are today in terms of the positive side [of the pandemic].”
Dr. Omer says the lesson he takes away from how things unfolded is that “at least in the US, it wasn’t a failure of science, because we had the scientific tools to control the outbreak. It was a failure of governance, and that’s a big lesson. Maybe we will find a way, a legislative or a policy way, such that we avoid these failures of governance [as seen] in the face of one of the most catastrophic events in recent history.”
According to Dr. Omer, reducing public health funding is one of the main failures of government that contributed to the severity of the 2020 pandemic.
“You can’t be penny wise and pound foolish in terms of investing in public health,” says Dr. Omer. “You can’t chronically starve public health infrastructure in states and cities at the federal level expect it to perform when you need it the most.”
While COVID-19 has undoubtedly been the biggest story in the life sciences in 2020, it was a busy and positive year in many other areas.
From drug approvals for rare diseases to innovative treatments involving mRNA technology, immunotherapies and gene therapies, and awarding of a Nobel Prize to the inventors of the gene-editing tool CRISPR, 2020 was a year of great activity and productivity despite the backdrop of the pandemic.
Here is a review of what 2020 served up.
COVID-19: The Pandemic Timeline and Journey

Save for a few countries such as Taiwan and South Korea that had been battle-tested by the related respiratory infection SARS more than a decade ago, the world was largely unprepared for a virus of pandemic proportions.
However, for virologists like Lori Frappier, PhD, professor in the department of molecular genetics at the University of Toronto, the SARS-CoV-2 outbreak was less of a surprise and more of an inevitability. She told Xtalks that, “Experts have always known that this kind of pandemic was a possibility, especially with coronaviruses. SARS-1 was a good warning. I think this pandemic will make the public more aware of the ability of viruses to mutate and jump from animals to humans.”
After being identified in December 2019 in Wuhan, Hubei in China, medical officials and scientists got to work to isolate and identify the novel virus that they observed to be causing a new respiratory illness. The illness was later named coronavirus disease 2019 (COVID-19).
The novel coronavirus behind the pneumonia-like disease was first isolated by Chinese researchers in early January and its genomic sequence was released on January 10. The virus was initially called 2019-novel coronavirus (2019-nCoV) but its name was later changed to severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2).
Shortly thereafter, the first case of COVID-19 was identified in the US on January 20. Researchers isolated the virus from nasopharyngeal and oropharyngeal samples from the infected patient and characterized its sequence and replication properties.
In Canada, researchers at the University of Toronto and McMaster University collaborated to isolate SARS-CoV-2 from two of the first COVID-19 patients in the country in January. Virologists Drs Samira Mubareka, Robert Kozak and Arinjay Banerjee led the team of researchers who are now working to develop better diagnostic tests, treatments and vaccines for the infection, as well as gain further understanding of the virus’ biology, evolution and clinical management.
Pandemic Efforts
Two months after the first cases in North America, COVID-19 was declared a pandemic by the World Health Organization (WHO) on March 11, 2020, with the rest being history in the making. And what a time it has been since.
In a span of just ten months, significant headway has been made in understanding the virology of SARS-CoV-2 — for example, we now know that it is not rapidly mutating despite two different variants having been found in Spain in the summer — as well as designing new drugs and re-purposing existing ones to treat the infection, while debunking the malaria drug hydroxychloroquine as a miracle COVID-19 cure.
In addition to pharma and biotech companies who zealously began working to develop COVID-19 drugs and vaccines, companies from areas outside of science and medicine also stepped up to help during the pandemic. Distilleries and breweries used their production facilities to make hand sanitizers while car manufacturers such as GM, Ford and Tesla, as well as vacuum maker Dyson, pledged to convert their assembly lines to make ventilators.
Clinicians also started to learn how to better manage the disease. For example, prone positioning — where patients in serious condition and on ventilators are placed on their stomach to help them breathe better — has been shown to have significant benefit and improved outcomes. Compared to the supine position (lying on the back, face up), the prone position results in less lung compression (from the heart and abdominal organs), which improves lung function, and can also improve air and blood flow as well as heart function, helping wean patients off ventilators quicker and more efficiently. Proning in voluntary, awake COVID-19 patients at risk of worsening respiratory status is also being studied now.
Drugs, Testing and Vaccines
Amidst the vaccine rush, there has also been a drive for finding old and new drugs to treat COVID-19. These include drugs that directly target the virus, to steroids and anti-inflammatories that help manage cytokine storm.
Dr. Frappier studies the Epstein-Barr virus (EBV) and said that while “EBV can hide from the immune system, coronaviruses can’t.” This makes the latter potentially easier to target with drugs and vaccines. She also explains that, “EBV has a persistent lifetime infection mode which SARS-CoV-2 doesn’t have.”
SARS-CoV-2 is characterized by an outer surface speckled with proteins called ‘spike’ that give it a crown-like appearance; hence the name “corona,” which is the Latin word for crown. The spike protein binds to ACE2 receptors on host cells — this interaction mediates the entry of the virus into cells. Most antibody drugs and vaccines have been developed to target parts of the spike protein. However, other components of the virus are also being studied and could serve as potential targets as well.
Speaking to Xtalks, Kevin Coombs, PhD, virologist and professor in the department of medical microbiology at the University of Manitoba, explains that the “immune system has two arms — humoral, which are the antibodies we all hear about; and cell-mediated, which is controlled by T cells. The spike protein is a natural target for both but is a little more important in humoral immunity.”
When asked about other potentially important parts of the virus, Dr. Coombs says, “Another SARS-CoV-2 protein that might be worth considering is the N (nucleocapsid) protein. This is an internal protein in the virus, made in large quantities, so is not that important for humoral immunity, but it is a good target for cell-mediated immunity.”
Drug Approvals
So far, the FDA has approved a handful of drugs for the treatment of COVID-19. These include remdesivir, baricitinib, dexamethasone and three antibody drugs.
While there were high hopes for Gilead Sciences’ experimental anti-viral remdesivir, it has shown minimal benefit despite being able to shorten recovery times in hospitalized adults with COVID-19. As of November, the WHO has discouraged the use of the drug as the agency’s review of data from four different clinical trials yielded no worthy effect on mortality, need for mechanical ventilation, time to clinical improvement or any other important patient outcomes. However, to potentially improve its effects, Eli Lilly’s anti-inflammatory JAK inhibitor baricitinib was approved by the FDA in October for use in combination with remdesivir.
On the other hand, the corticosteroid dexamethasone became the first drug shown to lower mortality in seriously ill COVID-19 patients, reducing deaths by as much as one-third in mechanically ventilated patients and one-fifth in patients only requiring oxygen support.
Antibody drugs were also developed as targeted COVID-19 treatments. Bamlanivimab, an antibody drug against SARS-CoV-2 designed via a partnership between Eli Lilly and AbCellera, as well as a dual antibody cocktail (casirivimab and imdevimab) from Regeneron, won EUAs from the FDA in November.
Diagnostics
With testing shortages posing to be an issue at the beginning of the pandemic, many companies were also racing to develop COVID-19 diagnostic tests. PCR-based tests continue to be the gold standard for detection of SARS-CoV-2.
With samples being delivered to central lab facilities for PCR analysis, the process could take up to a week. However, now, test results can be delivered within a day or two thanks to better coordination, ramped up production of test supplies as well as development of rapid tests.
Several saliva-based tests also received FDA approvals and authorizations, making sample collection easier and less invasive compared with traditional nasopharyngeal swabbing. Among these are the SalivaDirect test that the NBA used to routinely test its players, as well as tests by Phosphorus Diagnostics LLC and RUCDR Infinite Biologics, a unit at the Rutgers Clinical Genomics Laboratory at Rutgers University.
Innovations
COVID-19 fostered a number of innovations, ranging from plant-based vaccines being developed by Quebec-based company Medicago, to creating antibody drugs using lama antibodies.
The two FDA-authorized COVID-19 vaccines themselves are based on novel mRNA technologies that have not been employed for any approved vaccine for any other disease to-date. The pandemic helped accelerate the development of mRNA vaccines in record time.
While this was an exciting development, some still remain cautiously optimistic. Dr. Omer says, “Addressing an acute need in a pandemic and leveraging the best and latest approach to do that will, by definition, be new. So how do you deal with that and how do you mitigate some of the circumstances that arise when cutting edge technology is used?”
Dr. Omer maintains that the solution to this challenge is something that’s already in practice by developers of the mRNA vaccines.
“What you do is you insist that a proper clinical development program is followed and that has been followed with trial data of tens of thousands of people,” says Dr. Omer.
Vaccines
To round out the year on a positive note, major COVID-19 vaccine developers began posting results of their candidate vaccines from Phase III trials in November, the majority with positive efficacy data. Authorizations/approvals, vaccine shipments and the world’s first inoculations followed shortly thereafter.
Speaking to Xtalks about the effectiveness of the vaccines, Dr. Coombs says, “Like with influenza vaccines, the anti-SARS-CoV-2 vaccines should be effective against the virus they were designed against. However, as the virus evolves, the vaccine may lose its effectiveness. Thus, just like with the flu, we may need a different vaccine if the virus changes enough that the original vaccine is no longer effective.”
Germany’s BioNTech, in partnership with Pfizer, has led the way on the vaccine front with a demonstrated 95 percent efficacy for their mRNA vaccine, followed by Moderna at 94 percent and AstraZeneca and the University of Oxford’s collaborative viral vector vaccine showing an average efficacy of 90 percent. The AstraZeneca/Oxford vaccine results were a bit odd as a half dose of the vaccine ended up showing greater efficacy than the full two-dose regimen (due to an apparent scheduling mix-up); these results are currently under further evaluation.
BioNTech/Pfizer’s vaccine became the world’s first approved COVID-19 vaccine, with UK regulators becoming the first to authorize it for emergency use, followed by Health Canada, which fully approved it. Then, the US FDA followed suit by granting it emergency use authorization (EUA) in mid-December. Saudi Arabia also approved the vaccine, while Mexico has given it emergency use authorization.
Moderna’s vaccine received US authorization and approval in Canada later in December.
Vaccinations for priority individuals — namely seniors and healthcare workers in long-term care (LTC) homes — have begun in the US and Canada, having first kicked off in the UK where the inaugural shot was administered to a 90-year-old woman in Coventry.
Vaccine Considerations
The vaccine has no safety concerns, nor have any serious adverse events related to the immunization been reported thus far. However, regulators are warning people with allergies to polyethylene glycol (PEG), an excipient in Pfizer’s vaccine, not to get vaccinated just yet. In addition, although pregnant and breastfeeding women have been given vaccines for decades, some have raised concern that the vaccine was not tested in this population (although there are plans for it, and Moderna is also conducting fertility studies in mice).
As such, pregnant and breastfeeding women can choose whether they want to get the vaccine. In fact, the American College of Obstetricians and Gynecologists (ACOG) recommends that COVID-19 vaccines should not be withheld from pregnant or lactating women who meet criteria for vaccination based on the Advisory Committee on Immunization Practices (ACIP)-recommended priority groups.
Other vaccines made by companies including CanSino Biologics in China, the State Research Center of Virology and Biotechnology VECTOR Institute and the Gamaleya Research Institute (part of Russia’s Ministry of Health) have received early or limited use authorizations in those countries.
To ensure the equitable distribution of vaccines globally, 64 high-income countries have come together in the COVAX facility project led by Gavi, the vaccine alliance. As part of the initiative, countries will pool together resources to prevent any potential disparities in access to the COVID-19 vaccines.
Dr. Omer says, “it would be a tragedy if the disparities in resources translate into disparities to access to vaccines. But there are glimmers of hope — the COVAX advanced market commitment provides the mechanism for countries to access, at least for the first 20 percent of their population, a pretty significant supply of vaccines.”
He also says that the world has options. “There was good news from Chinese vaccines and the Serum Institute of India is on the course of producing the AstraZeneca/Oxford vaccine, so it is not a zero-sum game. The only vaccine available is not the one produced in Western countries. The restraint on vaccine doses may be more than the restraint on dollars. It’s not a given that we have to be in a highly inequitable vaccine distribution scenario come middle of 2021.”
An End in Sight
Going forward in the COVID-19 battle, virologists and epidemiologists say it is difficult to predict the course of the virus.
When asked by Xtalks whether SARS-CoV-2 could become a seasonal virus, a latent/chronic virus or be totally eradicated, Dr. Frappier said, “Coronaviruses are seasonal to some degree, in that there are more infections in the winter months. This is thought to be part of the issue now with the high spread of the virus in many countries. Coronaviruses have no latent mode of infection so it would not be expected that SARS-CoV-2 would set up a chronic infection. It is likely that the virus will develop mutations and these could make it less virulent.”
And in terms of seeing an end to the pandemic, Dr. Omer says, “Next year will be the story of reversing the course of this pandemic.”
While the vaccines will eventually help end COVID-19, he said “it won’t be an on or off switch. It will be like a dimmer — if we reach a certain level of vaccination, we will reach a new normal. Not a dystopian new normal, but society will make adjustments so that its functions come back.”
He explains that things will kick off a little bit slowly initially in terms of the visible impact of vaccines, but that “if the priority schemes are followed that prioritize the most vulnerable in terms of morbidity and mortality first, rather than trying to break transmission — because you need a certain critical mass of vaccines to have an impact on transmission — you will see an earlier impact on mortality, which will go down before infection goes down.”
And this would be a big win as COVID-19 mortality rates are currently hovering at around 3,000 deaths per day in the US.
“Because LTC residents and elderly get vaccinated first, you will start seeing a pretty substantial impact on mortality before transmission.,” says Omer. “But by late spring, [and into] summer, you will start seeing an impact on transmission as well. Around that time the weather will warm up which is favorable [in terms of seasonal effects of the virus] and vaccines will also ramp up.
“It’s hard to say when the inflection point will come; it could be in the fall or early winter. But nevertheless, 2021 into 2022 will be the time.”
CRISPR Gene Editing Inventors Win Nobel Prize
In non-pandemic news, it was a big year for CRISPR, the reigning genetic technology in molecular biology since it first debuted in 2012. Dubbed “genetic scissors” that can rewrite the code of life by this year’s Nobel Prize committee, the CRISPR/Cas9 gene editing system not only conferred on its inventors the Nobel Prize in Chemistry this year, but we also got to see some of the first results from clinical trials of CRISPR/Cas9-based gene therapies. The first CRISPR/Cas9 gene therapy was also directly administered to a patient through direct injection into the body (as opposed to modifying a patient’s cells with it and injecting them back).
In just under a decade, Jennifer A. Doudna, PhD, of the University of California, Berkeley and Emmanuelle Charpentier, PhD, of the Max Planck Unit for the Science of Pathogens Institute for Infection Biology, revolutionized genetic research by helping to identify a natural gene editing mechanism in bacteria. By re-tooling it, they were able to develop CRISPR/Cas9, a precision gene editing technology that can be used in any living organism. Owing to its precision, speed, versatility and ease of use, it was quickly adopted by thousands of labs worldwide.
The female scientist duo became the first women to be jointly awarded a Nobel Prize, making it a winning moment for women in science.
In their groundbreaking 2012 paper, Dr. Doudna and Dr. Charpentier demonstrated that the CRISPR/Cas system functions to provide adaptive immunity against foreign DNA in prokaryotes such as bacteria and archaea. When a virus infects bacteria (called a bacteriophage), parts of its genetic material can get incorporated into the genome of the bacterium as sequences called clustered regularly interspaced short palindromic repeats (CRISPR).
These sequences become a part of the prokaryote’s antiviral defense system, serving as genetic antibodies to detect and destroy foreign DNA from similar bacteriophages during subsequent infections. The system induces silencing of the foreign genetic material through RNA interference that involves pairing of CRISPR RNAs (crRNAs) with trans-activating crRNA (tracrRNA) — the RNA duplex then directs the CRISPR-associated protein Cas9 to cleave target DNA. The system can be used to precisely edit out portions of DNA flanked by CRISPR sequences.
As with any gene modifying approach, there are ethical concerns over its use for germline genome editing and manipulation of embryos, something that Dr. Doudna has been quite vocal about. The technology could get into the wrong hands and be used for more sinister purposes such as eugenics.
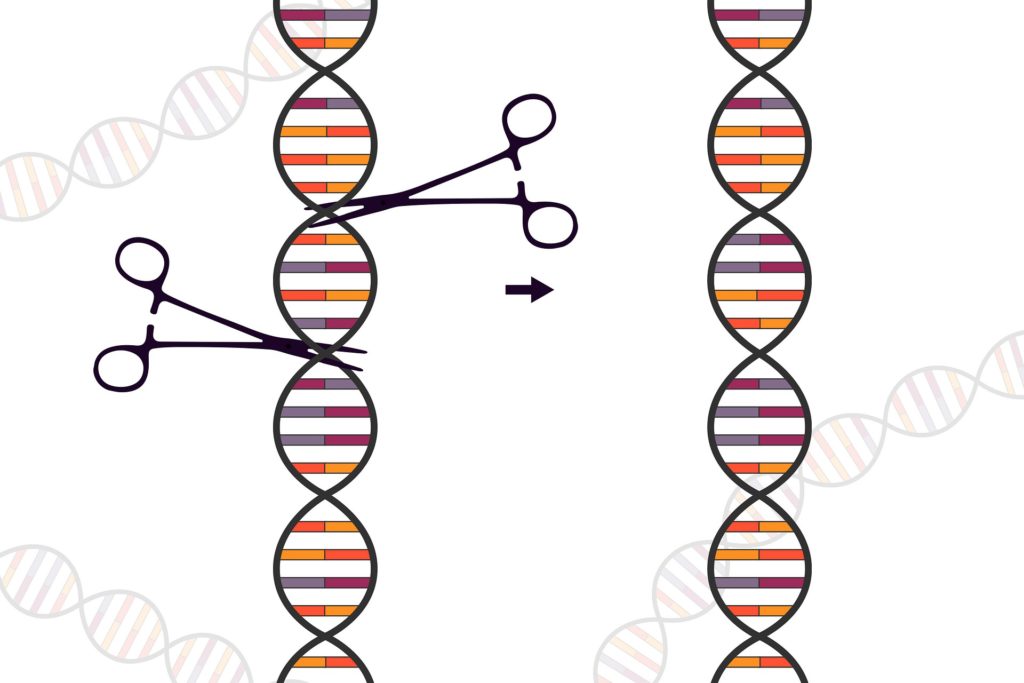
CRISPR Therapeutics
CRISPR is currently being evaluated therapeutically in a range of diseases and conditions, including muscular dystrophy, eye conditions, blood disorders and cancers. As a therapeutic, the CRISPR/Cas9 system is employed in target cells of interest — usually derived from patients — and then injected back into the individual.
This year, several promising studies showed the safety and efficacy of editing disease-causing genes in humans using CRISPR/Cas9 in various disease indications.
Clinical trials for a CRISPR-Cas9-based gene therapy targeting the BCL11A gene in blood disorders such as transfusion-dependent β-thalassemia (TDT) and sickle cell disease (SCD) have demonstrated effective, durable results. Both the diseases are characterized by compromised γ-globin expression and fetal hemoglobin due to enhanced activity of the BCL11A transcriptional repressor.
In a study led by Antonis Kattamis, MD, chief, division of pediatric hematology-oncology at the University of Athens in Greece, a patient with TDT and a patient with SCD were both treated with the CRISPR/Cas9-BCL11A targeted therapy, and more than a year later, both patients continue to maintain high levels of allelic editing in bone marrow and blood, pancellular increases in fetal hemoglobin and transfusion independence. The study was published this month in the New England Journal of Medicine.
Dr. Kattamis told Xtalks that, “the experience so far is of long-lasting effect, with the first patient reaching almost two years post infusion and doing well.”
He also explains that the CRISPR-edited cells seem to have a “survival effect” or advantage, and are thus unlikely to be ‘outpopulated’ by unedited cells.
Dr. Kattamis says, “This could very well be the first proof of a very powerful technology, which can be customized to treat a plethora of hematological diseases and cancer.”
Victoria Gray was the first patient sickle cell disease patient treated with the CRISPR gene therapy. More than one year after the receiving the treatment, the 35-year-old is doing well and was even able to get on a plane for the first time in her life — previously the altitude change would have caused extreme pain, a severe side effect of the disease.
CRISPR gene editing therapy is also being evaluated in cancers, including non-small cell lung cancer (NSCLC). In a study published in Nature Medicine, Chinese investigators led by Canadian-trained oncologist Tony Mok, MD, chairman, Li Shu Fan professor of clinical oncology at the Chinese University of Hong Kong, edited the immune checkpoint protein PD-1 using a CRISPR/Cas9 gene therapy. He and his team found that while the therapy was safe overall, there may be issues of off-target editing effects that require further evaluation and fine-tuning.
Big pharma has also stepped into the CRISPR arena as Pfizer released data from a Phase I trial for an investigational gene therapy being developed to treat Duchenne muscular dystrophy (DMD).
Preliminary data from the trial has shown that among nine boys (aged 6 to 12 years) with DMD who were intravenously administered a CRISPR therapeutic designed to target the dystrophin gene, the therapy was found to be well-tolerated with promising efficacy including durable and statistically significant clinical improvements. Prior to this, there have been many proof-of-concept preclinical studies showing the therapeutic utility of targeting the dystrophin gene in DMD.
In contrast to editing disease-causing patient cells, another group, for the first time, has directly injected a CRISPR/Cas9 gene therapy into a patient with a rare eye condition called Leber’s congenital amaurosis 10 (LCA10), which can cause blindness; it is the leading cause of vision loss in childhood. The first patient was dosed this year in the trial, which is evaluating a CRISPR gene therapy that corrects a mutation in the CEP290 gene that causes the disease.
Single-Cell Technologies
Single-cell technologies remained high-interest and in high-demand this year owing to continued improvements in resolution and the wide range of applications they can be used for. Interrogating the genome, transcriptome, proteome, metabolome and cell-cell interactions at the single-cell level offers valuable and unprecedented insights into potential variabilities in cellular characteristics and functions across different populations of cells in a tissue or tumor.
While technologies such as fluorescence-activated cell sorting (FACS) have allowed for profiling intracellular and cell surface markers in single cells, analyzing gene expression at the single cell level was previously not possible.
Traditionally, bulk samples are used for genomic profiling, which offers a broad overview of gene expression profiles across a milieu of different cells. For example, gold -standard PCR methods and sequencing technologies are typically performed using bulk samples. This leads to loss of critical information on the role of individual subpopulations within a sample.
Single-Cell RNA Sequencing
Next-generation sequencing technologies now allow for the sequencing of single cells at high resolution. Among these, single-cell RNA sequencing (scRNA-seq) is particularly useful for analyzing the transcriptome with respect to gene expression. Thousands of cells can be sequenced simultaneously, providing high-throughput data, allowing researchers to gain insights into differential gene expression across different cells in a sample.
The first step in most single-cell analysis techniques involves the isolation of individual cells. There are various methods for single cell isolation, some of which include dielectrophoretic digital sorting, enzymatic digestion, FACS, hydrodynamic traps, laser capture microdissection, manual picking, serial dilution and microfluidics.
In a study published in the journal Nature this year, researchers at MIT and Harvard used scRNA-seq to characterize molecular drivers and cellular dynamics in HIV infection. They performed scRNA-seq on peripheral blood mononuclear cells from untreated individuals before, and during the course of acute infection, to interrogate immune cell responses to treatment such as the expansion of potentially persistent natural killer cells.
An Xtalks webinar hosted by Genuity Science and 10x Genomics highlighted the utility of single-cell technology in the identification of immune cell populations in diseases such as hepatitis B. They explored immune profiling solutions for the characterization of multiple modalities from the same cell, such as gene expression and identification of T or B cell clonotypes based on high-resolution T cell receptor (TCR) or B cell receptor (BCR) expression analysis in response to various cytokines and drugs.
Single-cell technologies are particularly useful in oncology for the assessment of tumor heterogeneity. Many, if not most cancers, are highly heterogeneous with different cellular subpopulations that can evolve throughout the course of the disease and treatment. Therefore, it is important to identify disease-relevant clones and the evolution of potential new driver clones after treatment or at relapse. This can help guide more targeted treatments based on identification of disease driver mutations in specific cells that constitute a dominant clone in the disease.
Single-cell technologies will surely continue to make headlines in 2021 and beyond with the advent of higher-resolution techniques.
Immunotherapies
Immune-based therapies continued to gain traction this year with initiation of clinical trials and accrual of data for ongoing studies of various immunotherapies. Immunotherapies range from administration of immunomodulatory cytokines and growth factors to adoptive cell-based transfer therapies (like modified T cells) to blockade of checkpoint proteins.
Immunotherapies remain at the forefront of new oncology drugs, owing to initial successes in lymphomas and childhood leukemias like acute lymphoblastic leukemia (ALL). In addition to engineered chimeric antigen receptor (CAR) T cells now being a viable treatment option for several blood cancers, checkpoint inhibitors (targeting PD-1 and PD-L1 checkpoint proteins in T cells) are also used to treat dozens of cancers, including metastatic NSCLC and melanoma.
This year, the FDA granted approvals to 18 immunotherapies, the majority being for oncologic indications. This compared to 2019 in which about a dozen immunotherapies won FDA approvals.
In November, the FDA granted accelerated approval to naxitamab for high-risk neuroblastoma in bone or bone marrow. The approval was based on two open-label, single-arm trials that had 60 patients between the two studies. One trial showed an overall response rate of 45 percent with 38 percent in the other, and duration of response was greater or equal to 6 months in 30 and 23 percent of patients, respectively.
The FDA also approved the PD-1 checkpoint inhibitor Keytruda (pembrolizumab) for several cancers this year, including high-risk bladder cancer; adult and pediatric patients with unresectable or metastatic tumor mutational burden-high (TMB-H) solid tumors that have progressed following prior treatment; advanced breast cancer; cutaneous squamous cell carcinoma; and as a first-line treatment for microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR) colorectal cancer.
Looking Ahead to 2021
It has been quite the year for the life sciences, despite the pandemic being the focus for much of it. Perhaps the most important and relevant thing to be taken from the pandemic is the importance and utility of innovation in crisis situations. Case in point, mRNA vaccine technology — one of the newest and most sophisticated molecular technologies that was just about set to be tested in humans — provided a timely, innovative and effective solution.
The year 2021 will see much more of the pandemic, with continued management of record-breaking COVID-19 cases, and the emergence of new variants of SARS-CoV-2 in the UK to ponder alongside the rolling out of vaccines. Ending the pandemic will not only take vaccines, but also sustained pandemic measures and not letting our guards down.
The upcoming year will also see growth in many research areas such as AI-guided biological and drug discovery, as well as an increasing number of gene therapies entering clinical trials. Decentralized clinical trials will also be important as remote and virtual trials will continue to be a need both during and beyond the pandemic.
Despite the current events, the year is rounding out on an optimistic note with COVID-19 vaccines and continuing innovations to look forward to in the coming year.






Join or login to leave a comment
JOIN LOGIN