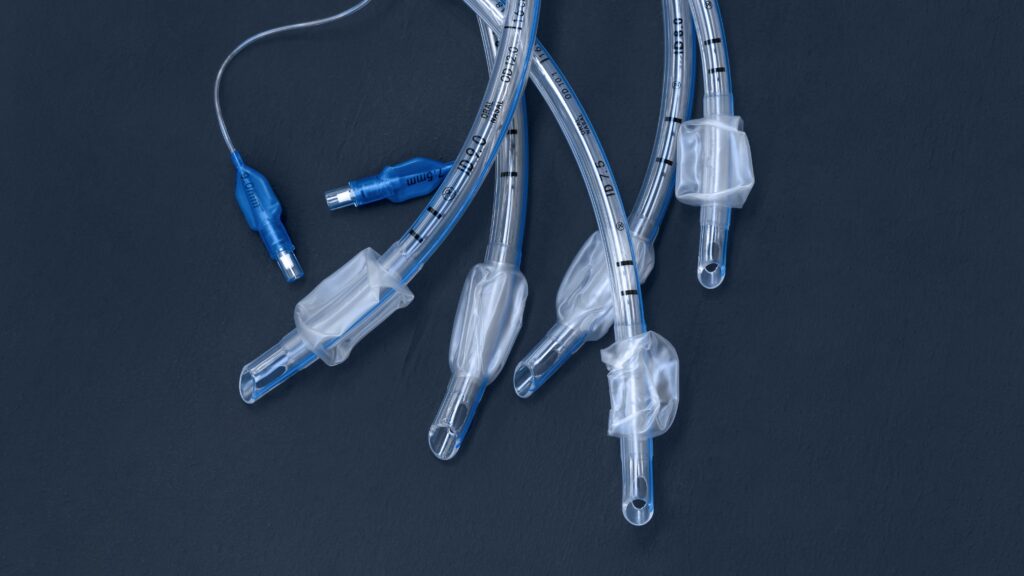
Smiths Medical has issued a recall of specific lots of its Intubation Oral/Nasal Endotracheal Tubes.
The FDA has classified the endotracheal tube recall as a Class I recall, the most serious type. This means use of the devices may cause serious injury or death.
The company is recalling the tubes because it discovered that some sizes may have a smaller diameter than expected. This could result in inadequate ventilation for the patient.
Smiths Medical recalled the tubes after quality testing revealed certain batches had incorrect sizes due to a manufacturing error. The company is a division of ICU Medical after its acquisition in 2022.
Endotracheal tubes help maintain an open airway to keep air flowing. They help ensure effective oxygen delivery during surgeries, emergencies and intensive care scenarios.
Clinicians insert them in a patient’s mouth (oral) and/or nose (nasal) during intubation to keep the airway open and support ventilation.
A reduced internal tube diameter can increase airway resistance and restrict airflow.
Underventilation can result in hypoxia (low oxygen levels) and respiratory distress. In severe cases, if not promptly identified and corrected, it can cause cardiac complications and death.
Related: Tack Endovascular System Recall: Philips Responds to Arterial Repair Issues
The recall includes a range of oral/nasal endotracheal tubes with specific lot numbers and size designations.
On February 13, 2025, Smiths Medical sent impacted customers an Urgent Medical Device Correction with recommended actions. These included checking inventory and stopping the use of affected products.
The company also advised customers to destroy all affected products or quarantine them if destruction was not possible.
It also asked people to complete a customer return form included with the letter, and to share the notification with potential users.
As part of the endotracheal tube recall, Smiths Medical is working closely with the FDA and healthcare providers to mitigate the risks and ensure rapid removal of the faulty products from circulation.
The FDA said healthcare providers and users can report any adverse events or quality issues related to the product to its MedWatch Safety Information and Adverse Event Reporting Program.
For details on affected product codes and return instructions, users can visit Smiths Medical’s website or contact their customer support team.
Smiths Medical has been having a tough month with recalls.
Just last week, it issued a Class I recall for its ProPort plastic implantable ports. Due to a manufacturing defect, the plastic port housing may separate from the port reservoir. Two injuries related to the defect have been reported.
Implantable ports are components of a venous access system that typically includes a plastic port, a silicone or polyurethane catheter, a connector, a Port-A-Cath access needle, a blunt needle and a vein pick.
ProPort systems are used for patients who need frequent venous access for injections, infusions or blood sampling.











Join or login to leave a comment
JOIN LOGIN