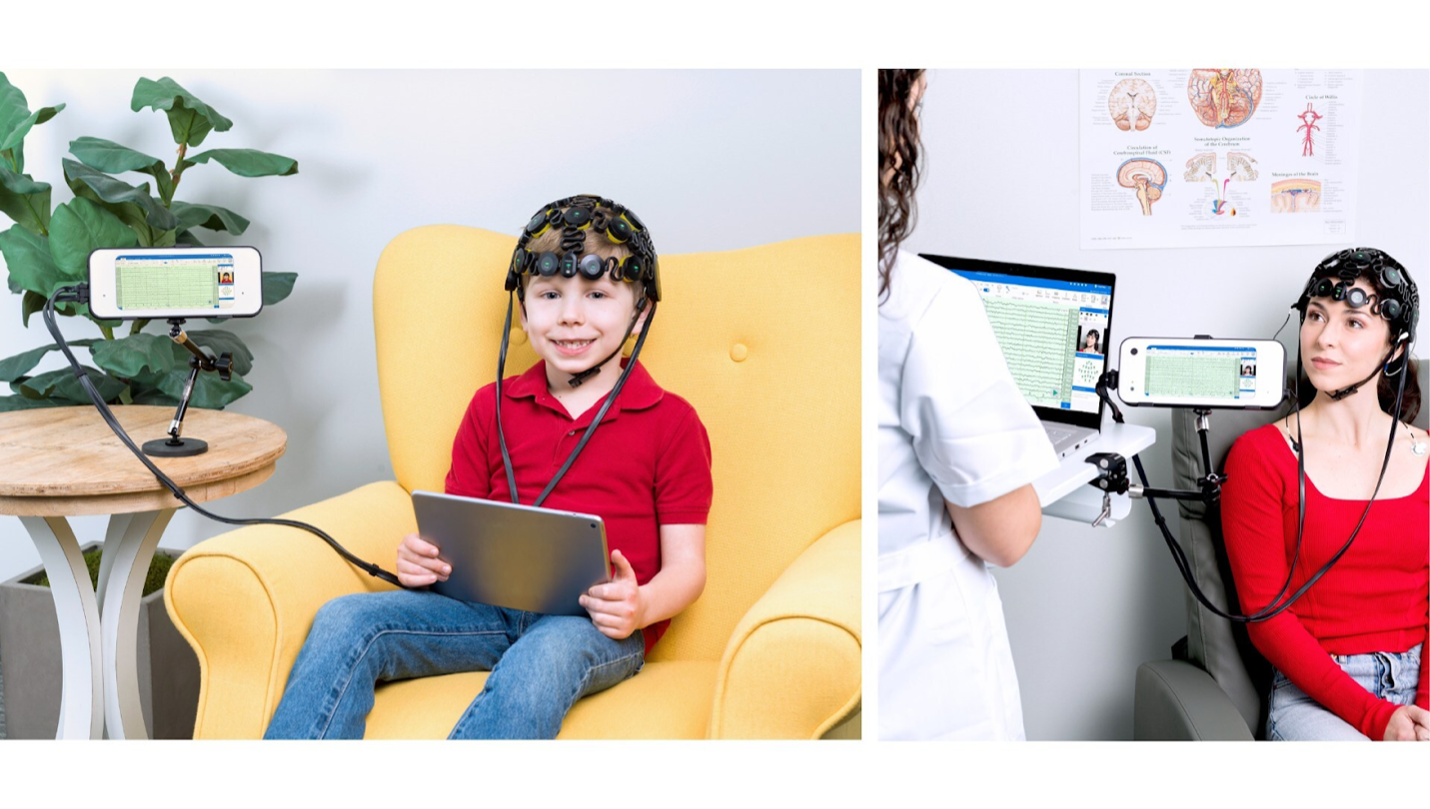
The FDA has approved a new once-monthly dosing schedule for Rybrevant Faspro (amivantamab and hyaluronidase-lpuj) for the first-line treatment of EGFR-mutated advanced non-small cell lung cancer (NSCLC), when used in combination with Lazcluze (lazertinib).
The monthly dosing option reduces how often patients need to come to the clinic. Under the updated label, patients may transition from the previously approved every-two-week subcutaneous dosing schedule to monthly administration as early as Week 5 of treatment.
Johnson & Johnson (J&J)’s Rybrevant Faspro is currently the only EGFR-targeted therapy with an FDA-approved once-monthly dosing option.
NSCLC is the most common form of lung cancer, and a subset of tumors is driven by activating EGFR mutations that promote cancer cell growth. Targeted therapies that inhibit EGFR signaling are a standard treatment approach for patients with these mutations.
Rybrevant Faspro is a subcutaneously administered formulation approved across all indications of intravenous Rybrevant (amivantamab-vmjw). The FDA previously approved Rybrevant Faspro in December 2025.
Amivantamab is a fully human bispecific antibody that targets EGFR and MET and can also engage the immune system. The formulation includes hyaluronidase to enable subcutaneous injection.
Support for the newly approved monthly dosing schedule comes from the PALOMA-2 trial, an open-label Phase II study evaluating first-line subcutaneous amivantamab in combination with Lazcluze, with or without chemotherapy in patients with EGFR-mutated advanced or metastatic NSCLC.
According to J&J, data presented at the 2025 World Conference on Lung Cancer showed that monthly dosing delivered outcomes consistent with the previously approved biweekly subcutaneous regimen. The primary endpoint of the study was objective response rate, assessed using RECIST v1.1 criteria. This means tumor response was measured using standardized imaging rules that define when tumors shrink, remain stable or grow.
The FDA decision was also supported by data showing that monthly dosing maintains similar levels of amivantamab in the body as the previously approved dosing schedules.
Development of the subcutaneous formulation was informed by the PALOMA-3 study, a randomized Phase III trial. The study evaluated whether subcutaneous and intravenous amivantamab resulted in similar drug levels in the body in patients with EGFR-mutated advanced NSCLC whose disease had progressed after osimertinib and chemotherapy.
The study met its co-primary pharmacokinetic endpoints based on measurements of amivantamab levels in the blood.
The updated dosing schedule is now reflected in the FDA-approved labeling for Rybrevant Faspro when used in combination with lazertinib for eligible patients with EGFR-mutated advanced NSCLC.
The company separately reported early-stage clinical data evaluating subcutaneous amivantamab and hyaluronidase-lpuj in recurrent or metastatic head and neck squamous cell carcinoma. The FDA granted Breakthrough Therapy designation to the investigational combination for this patient population.
In the Phase Ib/II OrigAMI-4 study, the investigational combination of subcutaneous amivantamab and a PD-1 inhibitor was evaluated as a first-line treatment in patients with recurrent or metastatic, HPV-unrelated head and neck squamous cell carcinoma whose tumors expressed PD-L1. Among treated patients, the study reported a confirmed overall response rate of 56%, with some patients experiencing complete tumor responses. Responses were observed within the first several months of treatment, and many patients remained on therapy at the time of data analysis.
Eli Lilly also reported Phase III trial results for selpercatinib, a targeted therapy being studied as adjuvant treatment in early-stage, RET fusion-positive NSCLC. In the LIBRETTO-432 study, selpercatinib was evaluated as an adjuvant treatment given after surgery or radiation intended to remove or control the cancer. Patients who received selpercatinib went longer without cancer recurrence or death compared with those who received placebo.
If you want your company to be featured on Xtalks.com, please email [email protected].











Join or login to leave a comment
JOIN LOGIN