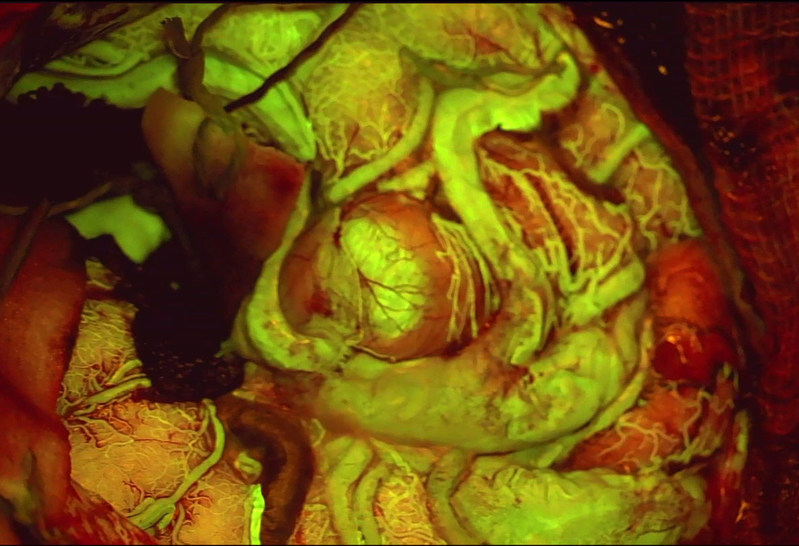
The US Food and Drug Administration (FDA) has approved Leica Microsystems’ FL560 fluorescence microscope filter to help surgeons better visualize cerebrovascular blood flow during neurosurgical procedures. The filter is designed to be used alongside the M530 OH6 neurosurgical microscope and the dye fluorescein, which is commonly used as a tracer in many medical applications.
According to Leica, the company is the first to be granted FDA approval for a medical device which uses fluorescence to monitor cerebrovascular blood flow during surgery. The contrast between the fluorescent blood flow and the surrounding tissue is designed to support surgeons’ decision-making when operating on a patient.
“Leica Microsystems has been a leader in advanced surgical visualization for decades,” said Markus Lusser, President Leica Microsystems. “I am proud that our team again was able to be the innovation leader by being the first company to achieve FDA clearance for cerebrovascular fluorescence imaging with fluorescein (FL560).”
The small blood vessels which supply oxygen and other nutrients to the brain can be difficult to visualize under white light alone. While near-infrared fluorescence has been used to add contrast, the image produced using this technique is only presented in black and white.
The Germany-based company’s FL560 filter allows for the simultaneous visualization of the brain’s vascular anatomy under white light, along with the fluorescent blood flow. According to Leica, the device is applicable to a variety of neurovascular surgical situations, including aneurysms and arteriovenous malformations (AVM), a rare condition in which abnormal blood vessels in the brain become intertwined.
Leica’s M530 OH6 neurosurgical microscope is also the first to allow three different types of fluorescence to be installed at once. Known as TriFluoro technology, this feature allows the surgeon to switch between different fluorescent modalities during a procedure.










Join or login to leave a comment
JOIN LOGIN