As the COVID-19 pandemic continues to grip the world with continued lockdowns and stay-at-home orders along with calls for greater testing, Canadian company Spartan Bioscience received approval from Health Canada this week for its fast and portable COVID-19 diagnostic test.
The test device is a hand-held DNA analyzer that detects the presence of viral genomic components through PCR-based technology in just under 30 minutes. The announcement comes as traditional test kits continue to be in short supply due to high demand.
Xtalks recently spoke to Paul Lem, MD, founder and CEO of Spartan Bioscience, leading up to the announcement this week, about the portable COVID-19 test device that the company has developed.
The device, known as the ‘Spartan Cube,’ is shaped as a cube, has a minimalist design and is the size of a large coffee cup, making for easy portability and use. In explaining how the test device works, Lem said “The work flow is that you collect a swab [sample] from a person, put the swab into [the] DNA test cartridge, put that cartridge in [the] device, and hit [the] ‘go’ [button], getting your results for COVID-19 in about 30 minutes. It’s super fast.”
Click ‘play’ to listen to the conversation
Fast turnaround times are of the essence and have been an issue of concern for COVID-19 testing as with current test procedures, results can take days and up to one week to be received.
Premised on its cutting-edge DNA analyzer technology, the test can process samples quickly and efficiently in just under half an hour. The technology includes specialized chemistry that allows its tests to be extremely rapid. Test results are provided in real-time, “going from sample to results in 30 minutes, which can be seen on an attached computer, tablet or smartphone,” said Lem.
Spartan Bioscience has 14 years of expertise in making fast and portable DNA analyzers for various applications, with nine issued patents and another six pending.
Some of Spartan’s commercial applications include a precision medicine test used for heart attack patients and an environmental water safety test that detects Legionella bacteria in water systems in buildings.
Spartan’s technology has regulatory approvals around the world including from the Food and Drug Administration (FDA) in the US. Several of Spartan’s commercial products have been used and validated by organizations like the Centers of Disease Control and Prevention (CDC), the Mayo Clinic and the New York State Department of Health.
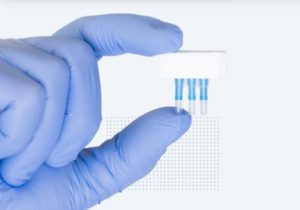
The COVID-19 test device is touted to produce highly reliable and accurate results “because we are taking the CDC’s published and validated COVID-19 test and simply putting that test onto our device,” said Lem.
As a portable device, the test is designed to be used in any type of a setting, making testing readily available when needed. Lem said that, “We really designed it to be used by non-technical people in places like small hospitals, remote communities, airports and border crossings.” Lem added that the goal is to essentially bring “portable DNA analyzers to everyone in making COVID-19 and other DNA testing accessible for everyone.”
Canadian Prime Minister Justin Trudeau had announced earlier last month that Spartan Bioscience was among three Canadian companies contracted to make COVID-19 tests, receiving fast-tracked federal funding for development and production. This was in response to addressing the gap between test kit shortages and increasing demand in the coming weeks.
Across Canada, the Government of Alberta announced that they have purchased $10 million CAD worth of the test, along with Ontario Premier Doug Ford whose government has purchased 900,000 tests.
“Everyday we are being solicited by other countries and corporations around the world that want to get access to our supply,” said Lem. “But we are trying to prioritize Canada because we are a Canadian company and proud to be Canadian [so] we are going to provide our test to Canadians first.”
Spartan Bioscience is working to scale up production of the devices so they can manufacture several hundred thousand of the test cartridges per week.
While the COVID-19 DNA test will not be available to the general public, Lem said that Spartan’s “ultimate vision is to drive the cost of these devices and tests down to the point where it will be like a blood glucose meter where it will eventually be in everyone’s home.”
When asked how testing should be done for the COVID-19 pandemic, Lem said that “For COVID-19 if you look at the countries that were able to bring the pandemic under control, it was countries like South Korea and a town in Italy and some places in China; one of the key components was that they tested everyone. Data from the CDC says up to 50% of people are actually asymptomatic so that’s why by mass testing, you can really bring a pandemic under control.”








Join or login to leave a comment
JOIN LOGIN