Takeda Pharmaceutical Company’s latest commercial for Entyvio highlights the struggle of patients who feel their lives are controlled by inflammatory bowel disease.
“Your head wants to do one thing, but your gut says, ‘Not today’,” says the narrator.
Entyvio (vedolizumab) is the Japanese pharmaceutical company’s most profitable GI drug, bringing in 201.0 billion Japanese yen ($1.8 million USD) in sales last year and driving 17.5 percent revenue growth. The monoclonal antibody stops T cells from homing in on the GI tract, reducing inflammation in the affected areas of the colon, rectum or small intestine.
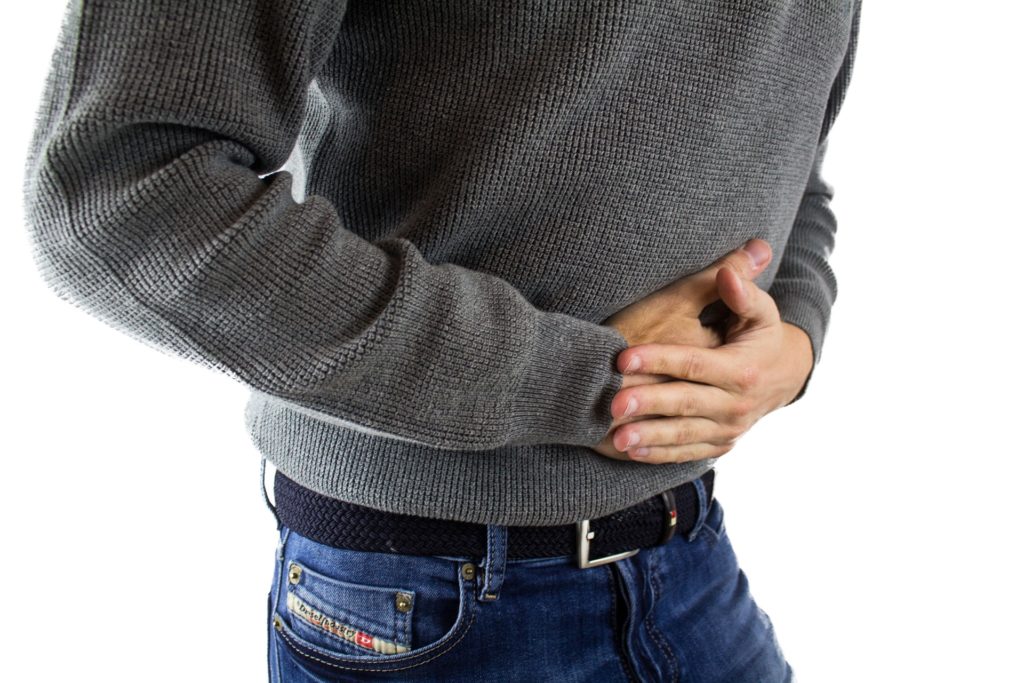
Moderate to severe ulcerative colitis or Crohn’s disease can affect a patient’s quality of life, captured in the new minute-long TV spot. A woman preparing for a camping trip is hindered by her symptoms — her day’s plan changed in a matter of minutes. A 2017 ad by Takeda sends a similar message, showing actors that feel “held back” by their symptoms.
According to the Centers for Disease Control and Prevention (CDC), about 3 million US adults were affected by inflammatory bowel disease including ulcerative colitis and Crohn’s disease. While there is no known cure (and no known cause either) patients can manage their gastrointestinal (GI) symptoms with corticosteroids or immunomodulators.
Entyvio has a large global footprint, approved to market in over 60 countries to date. Since its approval in the US and EU in 2014, Canada and followed suit in 2015 for ulcerative colitis and Crohn’s disease in 2016.
Most recently, Takeda Canada announced superior efficacy results in a study comparing Entyvio with AbbVie’s Humira (adalimumab). AbbVie also has a suite of direct-to-consumer TV ads for Humira.
The company is also seeking approval from the European Medicines Agency to sell a subcutaneous formulation of Entyvio, which is currently administered through IV. If approved, the company hopes to provide pre-filled syringes and pens as a more convenient option for patients.
The inflammatory bowel disease market is also shared by Pfizer’s Xeljanz (tofacitinib) and Johnson & Johnson’s Stelara (ustekinumab).










Join or login to leave a comment
JOIN LOGIN