
Director
Verrica Pharmaceuticals
Chief of Pediatric and Adolescent Dermatology
Rady Children’s Hospital-San Diego
This episode features an interview with Dr. Lawrence Eichenfield, chief of pediatric and adolescent dermatology at Rady Children’s Hospital-San Diego, and professor of dermatology and pediatrics and vice-chair of the department of dermatology at UC San Diego School of Medicine.
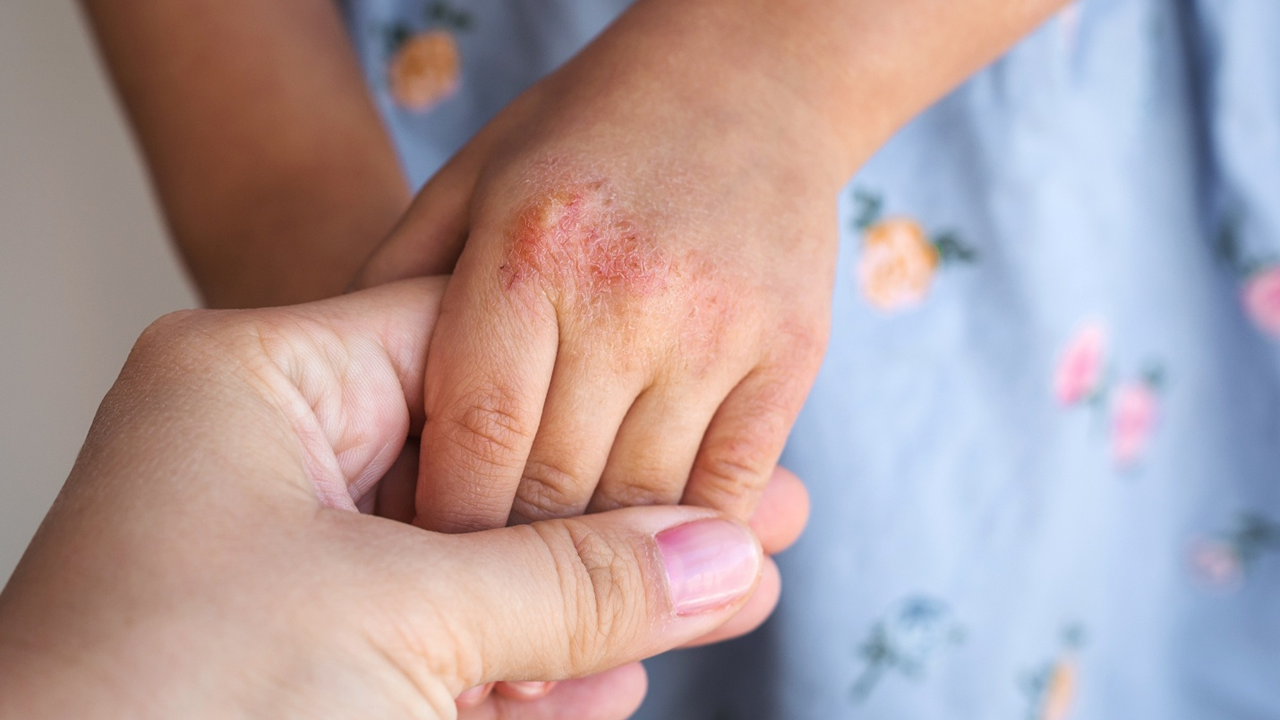
Dr. Eichenfield serves on Verrica Pharmaceuticals’ Board of Directors and spoke to Xtalks about the company’s recent approval of YCANTH (cantharidin) topical solution as the first FDA approved treatment for pediatric and adult patients with molluscum contagiosum, a highly contagious viral skin infection that primarily affects children. Dr. Eichenfield was also the principal investigator on clinical trials evaluating YCANTH.
Verrica Pharmaceuticals is a dermatology therapeutics company developing medications for skin diseases that require medical interventions.
Tune into this episode to learn more about YCANTH and its approval, as well as the changing landscape of dermatology, from Dr. Eichenfield.
The weekly podcast is available for streaming every Wednesday on Spotify, Apple Music and wherever you stream your podcasts.
Subscribe to the Xtalks Life Science Podcast to never miss a new episode.










Join or login to leave a comment
JOIN LOGIN