The way in which clinical trials are conducted is entering a new dimension as clinical trial operations are becoming more adaptable. The COVID-19 pandemic has caused a significant increase in the number of decentralized clinical trials (DCTs), which are performed away from primary clinical research study sites, as well as hybrid clinical trials (HCTs), which use a combination of centralized, site-based models and decentralized methods. In DCTs/HCTs, one or more components of the trial can be managed remotely from the patient’s home using wearable and remote monitoring technology.
“This focus has shifted to the concept of patient centric trials in which patient safety and convenience are prioritized without compromising data integrity and clinical trial endpoints,” says Dr. Richard Lee, MD, Senior Medical Director of the Medical Department at the global contract research organization (CRO) Medpace. “The COVID-19 pandemic certainly has pushed to the forefront of clinical trials, the digital evolution in clinical research. That was present prior but lagging behind innovation in technology.”
In a recent webinar, Dr. Lee described the current ecosystem of remote biosensor technology in clinical trials. In the same webinar, Dr. Salvatore Zabbatino, MD, Senior Director of Core Laboratories for Medpace spoke about selecting and integrating wearables into clinical trials. The webinar also included three case studies which used remote biosensor technology in DCTs, which were described by Mariel Fabro, Head of Products at Vivalink, a company which provides digital healthcare solutions for virtual patient care and DCTs.
Watch the on-demand webinar to gain valuable insights from a team of experts about the integration of wearable and remote monitoring technology in DCTs/HCTs.
What are Decentralized and Hybrid Clinical Trials?
DCTs are also called “virtual studies” or “direct-to-patient trials” because of the decreased reliance on visiting clinical research sites. DCTs are not limited to a geographical location because of the use of technology and services such as medical wearables, telemedicine and home visits, which allow data from patients to be captured remotely. Partially decentralized trials are also known as hybrid approaches or HCTs. Hybrid methods can involve patient visits at a designated clinical trial site for check ups, medical tests or procedures (e.g., MRI scan, endoscopy, etc.) as well as off-site data collection (e.g., teleconferencing, treatment administration, at home visits).
The Impact of the COVID-19 Pandemic on DCTs/HCTs
Before the COVID-19 pandemic, some virtual solutions such as remote coordinators, telemedicine and remote monitoring were being used for clinical trials. However, when the pandemic began, thousands of clinical trials around the globe experienced delays, disruptions or stopped completely. The US Food and Drug Administration (FDA) issued a guidance for industry, investigators and institutional review boards for the operation of clinical trials of medical products during the COVID-19 pandemic. The FDA guidance includes questions and answers about conducting virtual clinic visits, remote site monitoring visits and more. The European Medicines Agency (EMA) also issued a guidance on the management of clinical trials during the pandemic.
The interruption of clinical trials due to the COVID-19 pandemic has caused researchers to reconsider conventional methods for the conduction of clinical trials. This includes incorporating more DCTs/HCTs and using wearable and remote monitoring technology, among other methods. A 2020 industry survey showed that the COVID-19 pandemic has accelerated the acceptance of virtual solutions with 76 percent of Pharma, Medical Device, and CRO respondents report the use of decentralized trials; in addition, 38 percent reported that more than half of their trials are now decentralized. A clinical research site survey also shows a quick acceptance of virtual solutions since COVID-19 such as remote coordinators, telemedicine and remote monitoring since COVID-19.
“The digital evolution has now become what I call the digital revolution,” says Dr. Lee. “Indeed, the pandemic has ushered in a new era of patient-centric or personalized clinical trials. Certainly, that is here to stay.”
The Future of Decentralized and Hybrid Trials
DCTs are being increasingly conducted and have moved to the “center stage” due to the COVID-19 pandemic. An analysis of responses from 245 investigators in the US, UK and France predict an increased use of remote sensing technology and DCT procedures after the pandemic. For instance, most of the surveyed investigators expect telemedicine consultation, remote patient monitoring, remote site-initiation visits, electronic consent and in-home nurse visits will continue to be important DCT services in clinical trials after the pandemic.
The future of DCTs and HCTs certainly looks promising in a post-pandemic world. Dr. Lee says, “I think we can say with confidence that some form of virtual solutions will continue in clinical trials now and in the future.”
The Benefits and Challenges of Decentralized and Hybrid Trials
Clinical trials should be planned to consider the needs and wants of participants. DCTs are referred to as “direct-to-patient trials” because of the decreased reliance on a clinical research study site, which could be much more accessible and convenient for participants. The advantages of decentralized and hybrid clinical trials have been reviewed during the webinar and include the following:
- Patient centricity
- Trial access
- Adaptability of schedule of assessments
- Patient convenience and improved patient compliance
- Enrollment of a broader and more diverse study population
- Reduced burden to the site and patient
- Alignment with the FDA guidance that “safety is paramount”
- Possible cost savings with less site resourcing
However, it is important to keep in mind that fully decentralized trials have limitations and may not be possible for all clinical trial measurements or procedures. For instance, the collection of certain types of data or samples, such as diagnostic imaging data or biospecimen collection, may require an appointment at a healthcare facility. In addition, the administration of some complex treatments may need to be done at a healthcare center. For these reasons, HCT methods would be needed to allow participants to access health facilities, health care providers, diagnostic imaging centers and other resources.
Some challenges of DCTs and HCTs have been summarized during the webinar and include the following:
- Patient access to the internet
- Matching the right biosensor technology with the right digital endpoints
- Sites must be prepared to collect, integrate and harmonize prodigious amounts of data
- A learning curve with remote biosensor technologies for patients and sites
- Risk for protocol deviations with novel technology
- Quality of digital assessments taken remotely
- Scalability
- Increased cost of new technologies and software/application programming interfaces (APIs)
With increasing recognition of the value of telemedicine and technological advances, it is evident that DCT operations will develop and continue to be used in the future.
What are Wearable Biosensors and Why Use Them in DCTs?
Wearable biosensors are devices that can be worn as armbands, wristbands and in other ways to measure physiology parameters such as heart rate, respiration rate, skin temperature, oxygen saturation, actigraphy and more. The webinar presented devices and digital endpoints that can be used in decentralized studies. Some potential devices include blood glucose monitor, blood-pressure monitoring, ECG and actigraphy among others.
During the webinar, Dr. Zabbatino spoke about the incorporation of remote biosensors in clinical trials.
“From what used to be a want, or desire, from our sponsors, and perhaps even our regulators, now has almost become an expectation that we utilize these technologies, biosensors, remote visits and things along those lines,” says Dr. Zabbatino.
Incorporating wearable biosensors into decentralized and hybrid clinical trials has many advantages from the perspective of the patient, sites, sponsors and regulatory agencies, which are summarized in Figure 1.
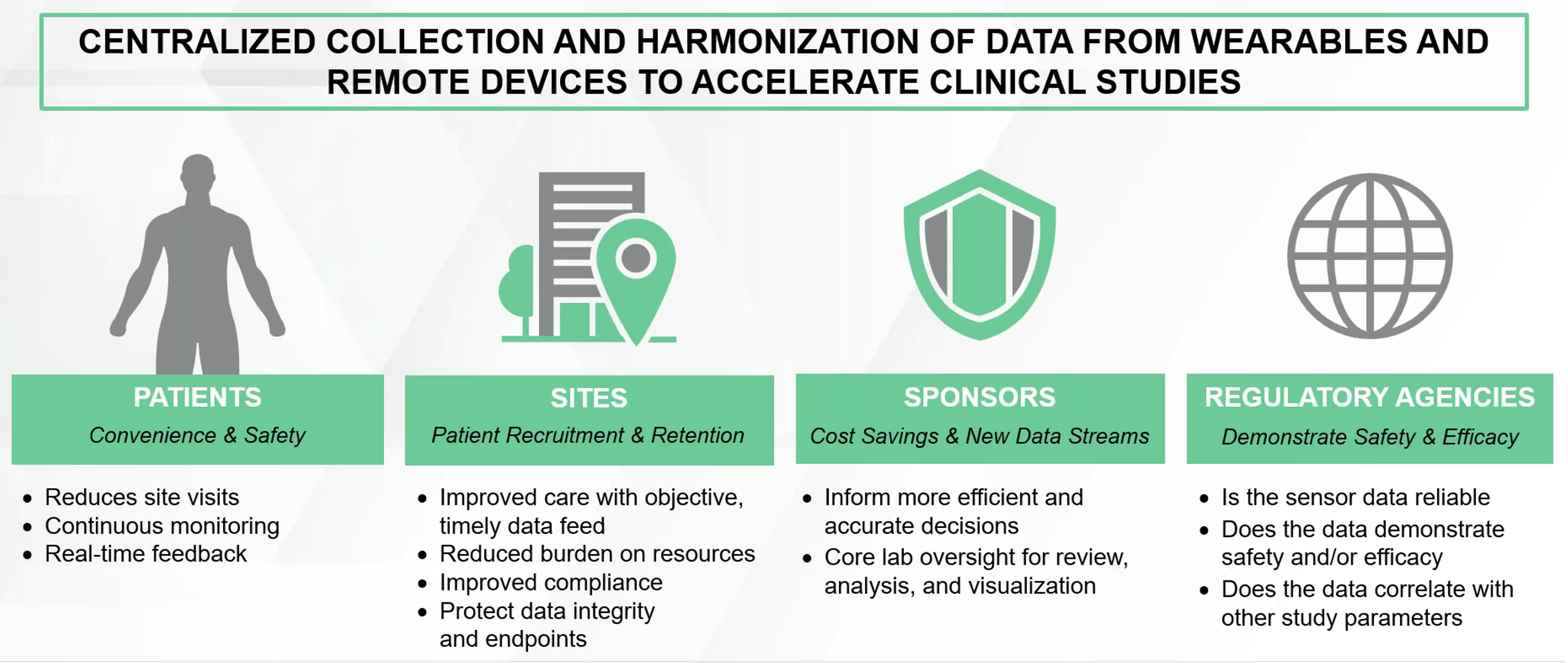
Figure 1. The advantages of using remote biosensors in clinical research.
CRO Medpace manages the operations of DCTs and HCTs and can incorporate remote biosensor technology within the framework of the clinical trial.
“Medpace has extensive experience in supporting DCTs and HCTs. For example, we’ve successfully conducted trials that leverage telemedicine, E Pro, E consent and various wearable monitors, such as rhythm monitors, actigraphy and continuous glucose monitoring,” says Dr. Lee. “Secondly, Medpace has keen experience integrating this data into the overall study and harmonizes it to present compelling cases to the regulators with the analysis of endpoints.”
Incorporating Wearable Biosensors in DCTs
To integrate a wearable biosensor in a decentralized or hybrid clinical trial, there are a number of factors to consider.
“We have to kind of keep in mind, what is the data that we’re looking for, is it what we really need? Just because we can monitor patients 24/7, 365, doesn’t always mean that we should,” says Dr. Zabbatino.
“As we decide on moving forward with picking these different biosensors, we have to make sure that the centers are available in the different regions that we’re going to be conducting these clinical trials, that the data from them is as validated, [and] that it has received approval, if necessary, from the different regulatory agencies in the different countries. We have to provide advice [and] logistics on how we’re going to get these devices to the site,” adds Dr. Zabbatino.
Patient privacy must be protected when using a wearable biosensor in a DCT and the data collected must be coming solely from the patient in the trial.
“We need to ensure that the data is being captured and stored in a protected manner, meeting all of those different regulatory requirements, and allowing us to visualize it in a way that allows us to act quickly for that. And we also need to integrate that with other study records,” explains Dr. Zabbatino.
An overview of wearable and device integration in clinical trials is shown in Figure 2.
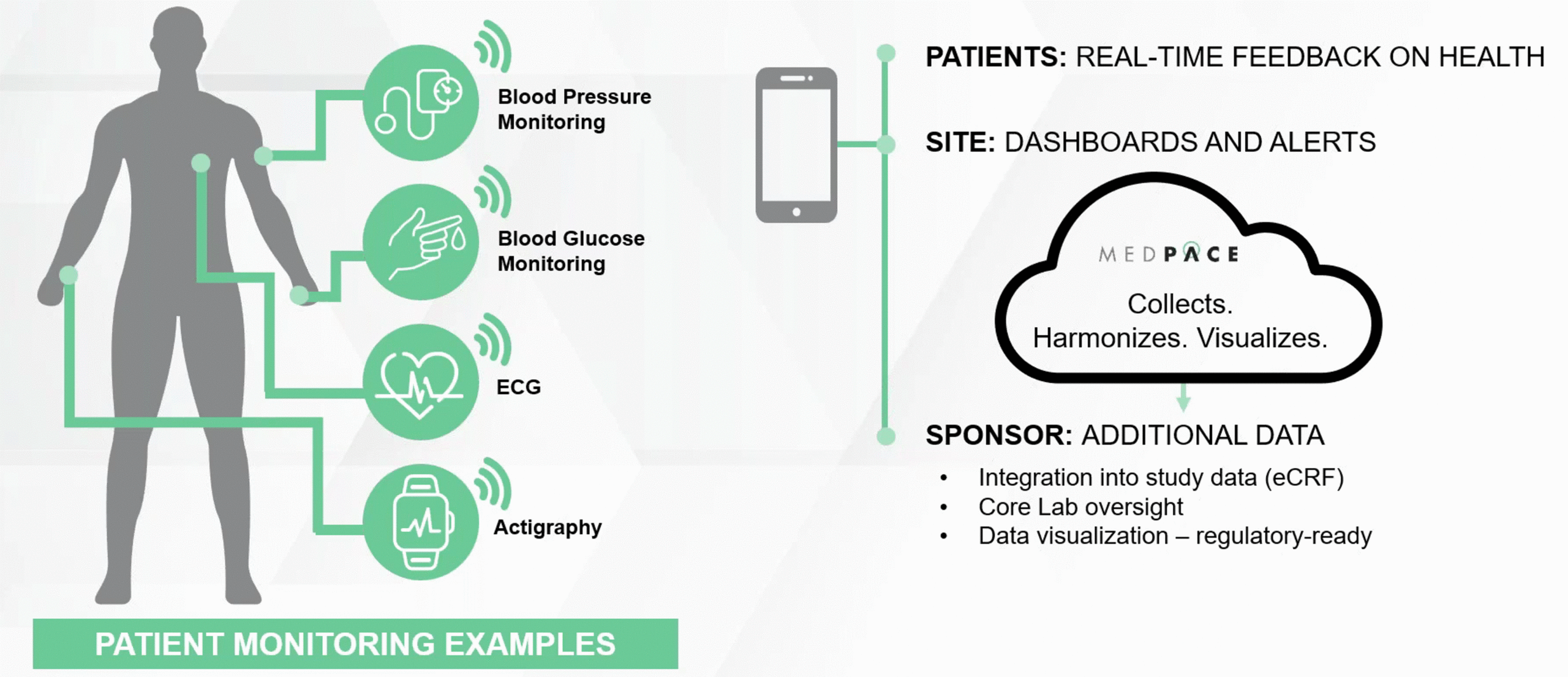
Figure 2. Patient monitoring examples and an overview of wearable and device integration in DCTs.
Real-World Case Studies Incorporating Wearable Biosensors in DCTs
The webinar featured three case studies in which wearable biosensors were used in DCTs. These case studies were made possible through a partnership between Medpace and Vivalink.
“With this partnership, we are leveraging expertise in biomedical signals and their relevance to therapeutic areas, as well as information technology infrastructure,” says Fabro. “In a classic approach here, we’re looking at body worn sensors which are continuously or episodically, transmitting data via wireless connections through things like mobile applications, which are very user friendly.”
Fabro explains that the data is then forwarded to data repositories, such as the cloud, for further analysis and appropriate visualization of the data via end use applications. The data could also be made to be compatible with conventional data analysis tools used in clinical research.
During the webinar, Fabro presented the following three case studies on the use of wearable biosensors in DCTs:
- Multi-parameter cardiopulmonary assessment for pulmonary hypertension
- Using remote continuous electrocardiogram (ECG) monitoring for digital biomarkers in atrial fibrillation
- Multi-vital biomarker in oncology for the early detection of neutropenic fever
For each case study, Fabro went into detail about the study objective, study type, digital measures, endpoints, key challenges and the solutions brought using wearable biosensors.
A key message from the webinar was that wearable biosensors should address accessibility for the patient, the clinical study team and the regulatory agencies. Remote biosensor technology can provide numerous benefits to the execution of decentralized and hybrid clinical trials.
To learn more about the incorporation of wearable biosensors in clinical trials, and the solutions wearable biosensors can bring to real-world case studies of DCTs, register to watch the free on-demand webinar by Medpace and Vivalink.
This article was created in collaboration with the sponsoring company and the Xtalks editorial team.




Join or login to leave a comment
JOIN LOGIN