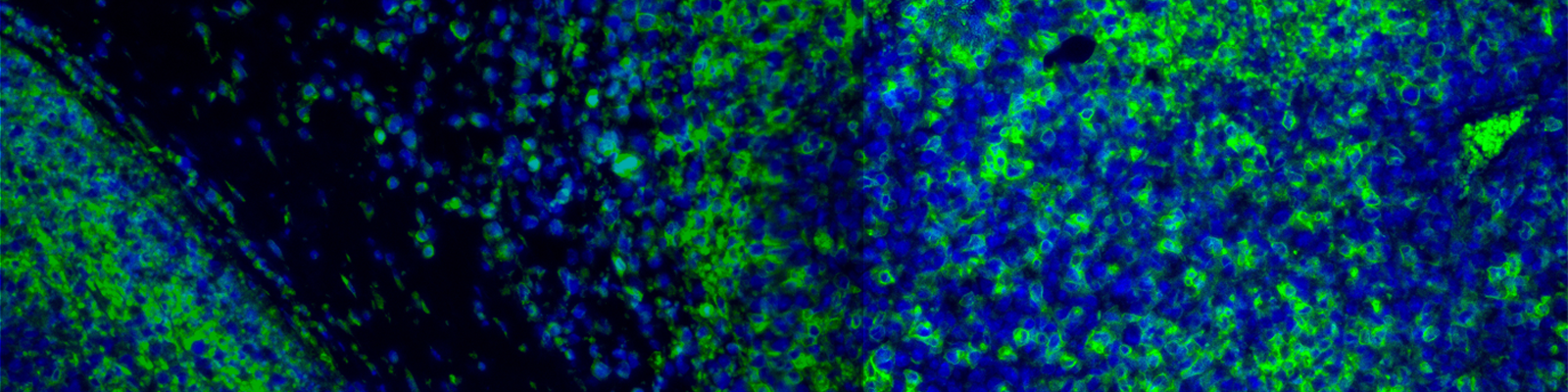
Immune infiltration in the tissue microenvironment is a hallmark of many disease indications, including cancer, autoimmune disease, degenerative conditions and others. Monitoring changes in immune heterogeneity according to disease state, or treatment with a drug is critical to understand disease etiology and predicting response to treatments.
In translational research, it is critical to develop assays that accurately measure biomarkers and the colocalization of immune cells within the context of the tissue microenvironment. Importantly, developing immunohistochemical biomarker assays that establish changes in pharmacodynamic relationships within the tissue context is important for measuring therapeutic response and predicting the likelihood of successful treatments in the clinic.
The multiple cell phenotypes of immune cells can be interrogated simultaneously on the same formalin-fixed paraffin-embedded (FFPE) section using multiplex immunohistochemistry (m-IHC). As such, multiplex immunofluorescence (m-IF) assays have become an established approach to measure immune biomarkers, from early discovery to exploratory endpoint testing in clinical trials. Despite the high utility of these assays in understanding mechanisms of disease, development and optimization is difficult due to the high complexity and need for deep domain and technical expertise, from staining to whole slide fluorescent imaging and analysis.
Register for this webinar to hear experts from Invicro review the current challenges using these approaches encountered in biomarker discovery and development programs. Specifically, you will learn how using a combination of multiplex immunostaining and multispectral image analysis approaches can uncover insights needed to accelerate the development of tailored therapies.
Speakers

Joseph Krueger, Ph.D. | VP of Research & Applications, Advanced Pathology Services, Invicro
Dr. Krueger serves as the Vice President of Research and Applications for Advanced Pathology Services at Invicro. In his role, Dr. Krueger is responsible for developing new biomarker applications to meet drug development needs, and provides scientific expertise needed to support sponsor-based projects across therapeutic areas. Prior to joining Invicro, Dr. Krueger was Chief Scientific Officer at Flagship Biosciences, Inc., where he led the R&D and Scientific efforts in applying novel quantitative tissue image analysis approaches to immuno-oncology and rare diseases to support drug and companion diagnostic development. Prior to Flagship Biosciences, he served in senior scientific roles in oncology drug development for OSI Pharmaceuticals in Boulder, CO and Pfizer in Andover, MA. Dr. Krueger completed his postdoctoral fellowship at The Scripps Research Institute and The University of California, San Diego after completing his Ph.D. in Cancer Biology at Wayne State University in Detroit, MI.

Amanda Bares, Ph.D. | Senior Scientist, Advanced Pathology Services, Invicro
Dr. Bares is a Senior Scientist and Study Director in Invicro’s Advanced Pathology Services group. As an expert in fluorescence microscopy, multiplexed assay development, and multiplexed imaging, she leads many projects across various therapeutic areas requiring advanced staining and imaging approaches. Prior to joining Invicro, Dr. Bares was an R&D Senior Scientist at Ultivue, a Cambridge-based startup developing pre-optimized multiplexed immunofluorescence kits for immuno-oncology applications. There, she played a key role in choosing the fluorescent dye panels used and established kit compatibility with a variety of commercial autostainers and whole slide scanners. During her Ph.D., Dr. Bares built a hyperspectral two-photon microscope capable of acquiring in vivo multiplexed images using spectral unmixing. She completed her Ph.D. in Biomedical Engineering at Cornell University in 2017 and her Bachelor of Science in Electrical Engineering at Montana State University in 2011.
Who Should Attend?
This webinar is recommended for pathologists and research scientists focused on drug discovery and development, which requires analysis of the immune infiltrates in tissue. By attending this webinar, attendees will better understand how domain and technical expertise is applied to design multiplex immunofluorescence assays to detect biomarkers and understand contextual relationships across tissue types.
What You Will Learn
In this webinar, attendees will learn about:
- Current challenges in drug development and how Invicro applies domain expertise to address them
- Limitations of conventional IHC methods and how multiplex fluorescent assays can be deployed to detect biomarkers and characterize immune cells in the tissue microenvironment
- Utility of novel research tools and methods developed by Invicro to answer specific biological questions
Xtalks Partner
Invicro, A Konica Minolta Company
Headquartered in Boston, MA, Invicro was founded in 2008 with the mission of improving the role and function of imaging in drug discovery and development across all therapeutic areas. Today, Invicro’s multi-disciplinary team provides solutions to pharmaceutical and biotech companies across all stages of the drug development pipeline (Phase 0-IV), imaging modalities and therapeutic areas, including neurology, oncology, and systemic and rare diseases. Invicro’s quantitative biomarker services, advanced analytics and AI tools, and clinical operational services are backed by Invicro’s industry-leading software informatics platforms, VivoQuant™ and iPACS®, as well as their pioneering IQ-Analytics Platform, which includes AmyloidIQ, TauIQ and DaTIQ.
As a CAP-accredited, CLIA-certified laboratory, Invicro offers a wide-range of anatomical and molecular pathology services including, core histology, multiplex immunohistochemistry and immunofluorescence, RNAScope® ISH, BaseScope™ ISH, pathologist interpretation, data management, image analysis and Quanticell™, a proprietary nanoparticle-based immunohistochemistry detection technology. Our pathology service laboratories support projects spanning biomarker discovery to clinical trials and companion diagnostics. As part of the Konica Minolta precision medicine organization and with their sister company Ambry Genetics, Invicro develops and leverages the latest approaches in quantitative biomarkers including imaging, quantitative pathology, and genomics. For more information, visit www.invicro.com.
You Must Login To Register for this Free Webinar
Already have an account? LOGIN HERE. If you don’t have an account you need to create a free account.
Create Account