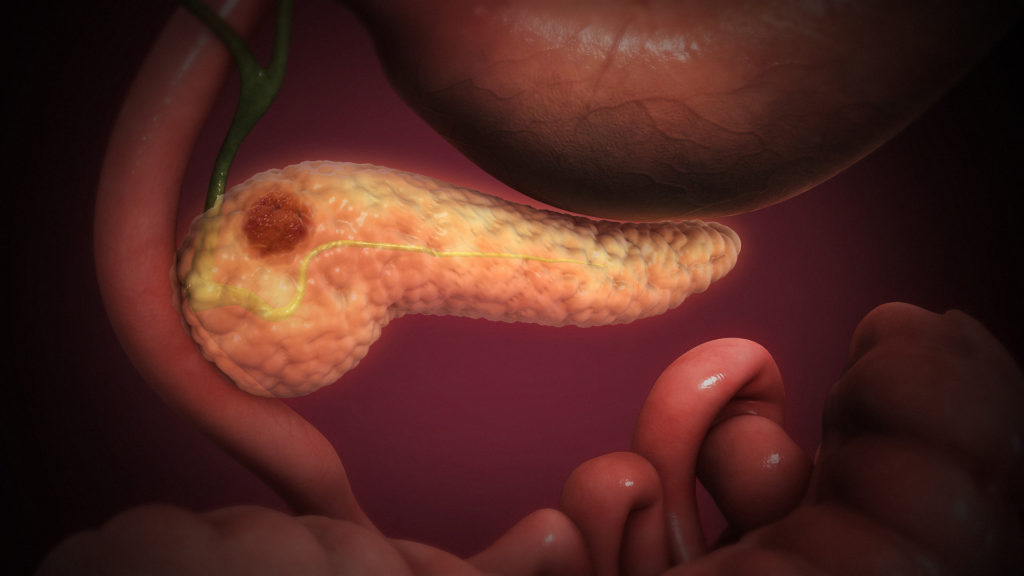
Compared to other cancer types, advances in pancreatic cancer treatment have been lagging behind. Over the last 40 years, one study found that the incidence and mortality rate of pancreatic cancer has been rising. The five-year survival rate for all stages of pancreatic cancer remains at about nine percent.
However, AngioDynamics, a New York-based biotech company hopes to change the outlook for pancreatic cancer patients. Following US Food and Drug Administration (FDA) approval, they have launched a new clinical trial testing their NanoKnife technology, a device that destroys cells through targeted electrical pulses, to help people with stage III pancreatic cancer.
The electrical pulses sent through the NanoKnife creates pores in the cell membrane, effectively killing the cancer cells. This form of ablation therapy is known as “irreversible electroporation”.
“Existing evidence that has accumulated over the last 10 years has shown that [irreversible electroporation] is an effective adjunctive treatment for patients with locally advanced pancreatic cancer,” said Dr. Robert Martin, Co-Principal Investigator of the DIRECT Study and Surgical Oncologist at the University of Louisville. “We anticipate that this trial will demonstrate how the NanoKnife System can be utilized to enhance the quality of life for patients with Stage III pancreatic cancer.”
The company hopes to establish up to 15 sites for their randomized controlled trials, with each site managing 500 participants. Additionally, they will set up 45 real-world evidence registries to support their clinical findings.
The NanoKnife’s regulatory journey began over a decade ago, receiving 510k clearance in 2008 for soft tissue ablation. Last January, NanoKnife gained breakthrough device designation to expand its indication to stage III pancreatic cancer, where the cancer might have spread to nearby lymph nodes. Now, the company got the go-ahead to begin clinical testing under an investigational device exemption (IDE), meaning they will have to use their clinical data to support a pre-market application.
The NanoKnife differs from the company’s other tissue ablation devices: StarBurst, a thermal-based tissue ablation system equipped with real-time monitoring and Solero, a device used to destroy tissues with microwaves. Each technique is considered minimally invasive, involving ultrasound or CT scan guidance plus laparoscopic surgery.
Another tissue ablation technique is known as cryoablation, where the tumor is destroyed by freezing it. Cryoablation paired with immunotherapy has been found to be effective at killing cancer cells at a lower cost.
Other companies have taken advantage of electrical current to target cancer cells. Instead of opening pores in the cell membrane, a device by Advanced Chemotherapy Technologies uses electrical currents to carry chemotherapy drugs directly into the tumor. The startup received a $1 million grant from the National Institutes of Health to begin clinical testing of their device in patients with pancreatic adenocarcinoma.
With these new therapies under clinical investigation, patient advocacy groups like the Pancreatic Cancer Action Network are encouraging people with the disease to start getting involved.
“The DIRECT study design demonstrates our commitment to provide an option that addresses a pressing unmet need for patients with Stage III pancreatic cancer,” said Jim Clemmer, President and Chief Executive Officer of AngioDynamics. “We are confident that our comprehensive approach, crafted through a constructive dialogue with the FDA, will generate meaningful data for clinicians, patients, payors and other stakeholders who are equally committed to fighting this disease, and we are dedicated to the idea that the standard of care for this deadly disease can—and should—improve.”









Join or login to leave a comment
JOIN LOGIN