Braeburn, a US pharmaceutical company specializing in the treatment of opioid use disorder, announced that their drug Brixadi (buprenorphine) has received approval from the US Food and Drug Administration (FDA). Brixadi is an extended-release injection intended for subcutaneous use to address moderate to severe opioid use disorder.
Brixadi is specifically indicated for patients who have already started treatment with a single dose of a transmucosal buprenorphine product (that enters across a mucous membrane) or are currently receiving treatment with a transmucosal buprenorphine-containing product.
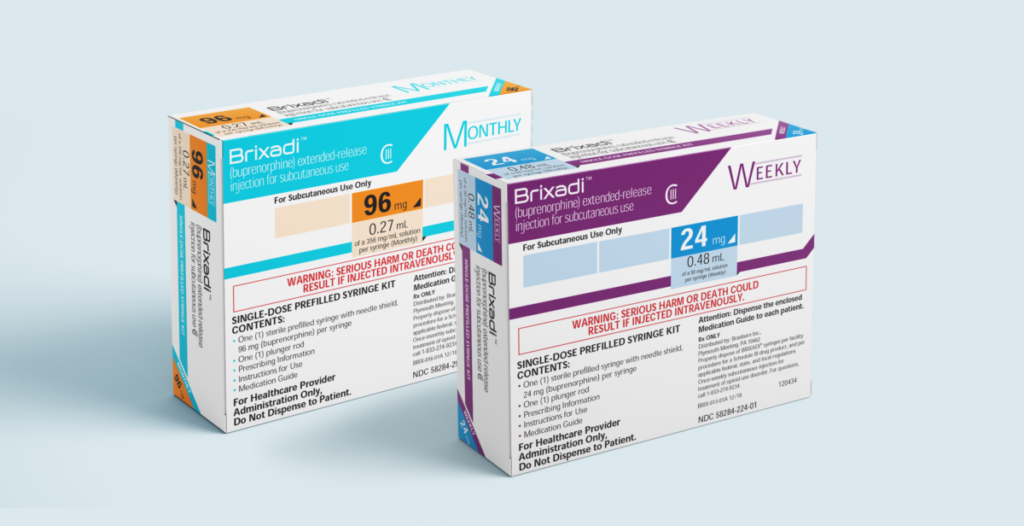
Brixadi is the first and only long-acting buprenorphine injectable available in both weekly and monthly doses.
Mike Derkacz, president and CEO of Braeburn, expressed enthusiasm regarding the FDA’s approval of Brixadi, stating, “Today’s FDA approval of Brixadi represents a major advancement in combating OUD [opioid use disorder]. Over the past three years, the US has witnessed a significant rise in opioid overdoses, which can be attributed in part to the economic and social challenges faced by all individuals, particularly those with OUD and those in recovery. The availability of additional medication options for OUD will aid healthcare providers in meeting the needs of their patients.”
Opioid overdose remains a major contributor to accidental deaths in the US, with a significant rise in fatalities observed in recent years. According to the Centers for Disease Control and Prevention (CDC), an estimated 107,622 drug overdose deaths occurred in 2021, reflecting an alarming increase of nearly 15 percent compared to the estimated 93,655 deaths in 2020.
Opioids are frequently prescribed for pain management due to their potent analgesic and sedative properties. These medications exert their effects by interacting with opioid receptors in the brain. Commonly used opioid drugs include morphine, fentanyl and tramadol; however, the misuse or overuse of these substances can lead to dire consequences.
Symptoms of opioid overdose typically manifest as pinpoint pupils, loss of consciousness and respiratory difficulties. It is essential to be aware of these signs and promptly seek medical assistance in suspected cases of opioid overdose to prevent further harm.
RELATED: Narcan Nasal Spray Becomes First OTC Naloxone Product
How Does Brixadi (Buprenorphine) Work?
Brixadi (buprenorphine) is an extended-release injection available as a sterile liquid. It comes in a single-dose, pre-filled syringe intended for subcutaneous injection only. The active medical ingredient in Brixadi, buprenorphine, acts as a partial agonist at the mu-opioid receptor and an antagonist at the kappa-opioid receptor. Its mechanism involves binding to the brain’s mu-opioid receptor, which helps block the subjective opioid drug-liking effects.
Brixadi is specifically formulated for extended-release delivery of buprenorphine. It is offered in two regimens: weekly and monthly. Brixadi (weekly) contains 50 mg/mL of buprenorphine base and should be administered every seven days. On the other hand, Brixadi (monthly) consists of 356 mg/mL of buprenorphine base and should be administered every 28 days.
Brixadi is formulated with FluidCrystal Injection Depot Technology. Following subcutaneous injection, Brixadi undergoes a spontaneous transformation from a low viscous solution to a nanostructured liquid-crystalline gel. This gel encapsulates buprenorphine and gradually releases it at a consistent rate as the depot biodegrades over a one-week or one-month period.
Due to its crystalline gel formulation, Brixadi is not to be administered intravenously because it could cause harm or death if injected into the veins.
Safety and Efficacy of Brixadi
The FDA’s approval of Brixadi is supported by a 24-week Phase III double-blind, clinical trial involving 428 adults with moderate to severe opioid use disorder. In this trial, patients were randomized to receive either Brixadi injections with placebo sublingual tablets or sublingual buprenorphine/naloxone (SL BPN/NX) tablets with placebo injections.
Weekly and monthly Brixadi met the primary endpoint of non-inferiority for responder rate versus daily SL BPN/NX (16.9 percent versus 14.0 percent, respectively; p<0.001). Additionally, Brixadi demonstrated superiority to SL BPN/NX based on the cumulative distribution function of the percentage of negative opioid assessments from Week 4 through Week 24 (p=0.004; secondary endpoint).
“A weekly and monthly buprenorphine injection with different dose options can align with clinical practice and patient care needs,” said Dr. Michelle Lofwall, professor of behavioral science and psychiatry at the University of Kentucky Center on Drug and Alcohol Research and primary investigator in the Phase III study.
Adverse reactions commonly associated with Brixadi administration were injection site pain, headache, constipation, nausea, injection site erythema (abnormal redness of skin), injection site pruritus (itchy skin), insomnia and urinary tract infection.
To ensure safe usage, Brixadi will be accessible exclusively through a restricted distribution program called the Brixadi Risk Evaluation and Mitigation Strategy (REMS) Program and will be administered solely by healthcare providers. The company did not list the price of Brixadi, but most patients will likely pay $10 or less for Brixadi.
Opvee (Nalmefene): A New Emergency Treatment for Opioid Overdose
Indivior, a global pharmaceutical company dedicated to developing medicines for the treatment of substance use disorders and serious mental illnesses, has recently announced the FDA approval of its new medication Opvee (nalmefene).
Opvee is a nasal spray designed for the emergency treatment of known or suspected opioid overdose. It contains nalmefene, an opioid receptor antagonist that rapidly reverses opioid-induced respiratory depression and has a long duration of action.
The approval of Opvee was supported by safety and pharmacokinetic studies. In a clinical trial involving 61 opioid-experienced, non-dependent subjects, it was found that the reversal of respiratory depression occurred within 2.5 to five minutes after administration of Opvee. Complete recovery of respiratory drive was demonstrated as early as five minutes after administering Opvee.
June 21, 2023, update: The title of this article was updated to reflect the fact that Brixadi is the first and only long-acting buprenorphine injectable that is available in both weekly and monthly doses.











Join or login to leave a comment
JOIN LOGIN