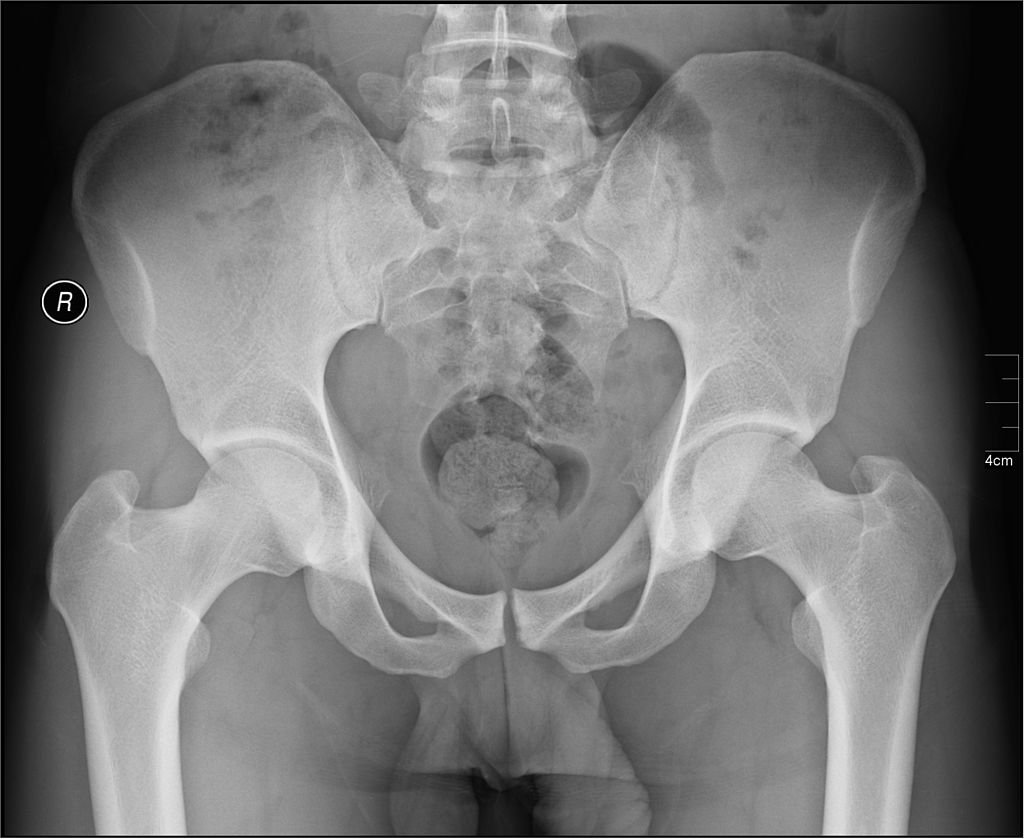
An independent data monitoring committee has recommended that Regeneron discontinue administering high doses of its investigational osteoarthritis drug fasinumab in its ongoing Phase III clinical trials. While the risk benefit profile for higher doses was unfavourable enough for the committee to make its recommendation, Regeneron will continue clinical development of the lower dose forms of fasinumab.
The drug is an anti-NGF antibody that Regeneron is currently studying in a number of indications, including osteoarthritis of the hip or knee and chronic back pain. The series of Phase III clinical trials have enrolled thousands of patients to date, however Regeneron will now need to alter its study protocol to eliminate the high-dose treatment arms.
“An independent Data Monitoring Committee monitoring the ongoing safety and efficacy of the fasinumab clinical trials recommended that the higher dose-regimens be discontinued based on the risk benefit assessment and that the program may continue with the lower dose-regimens of fasinumab,” said Regeneron’s first quarter 2018 earnings report. “The trials are being modified accordingly.”
Regeneron and Teva are collaborating on the clinical development of fasinumab in a deal that was inked back in 2016. Since then, the companies have faced a few setbacks including an FDA-mandated clinical hold on a Phase IIb study of the drug after one patient developed adjudicated arthropathy, a disease of the joints.
Johnson & Johnson and Pfizer also experienced similar setbacks with their development of anti-NGF antibody drugs, hinting at potentially deep-rooted problems with the once celebrated drug class. Clinical holds were often ordered after clinical trial participants required joint replacements.
While it’s unclear why drugs like fasinumab seem to increase the risk of joint damage when administered in high doses, some believe that their painkiller properties could push patients with arthritis to increase their activity level, thereby increasing the rate at which the cartilage breaks down.
But with the focus on the opioid epidemic showing no signs of slowing down, Regeneron seems intent on completing their clinical program for the non-opioid painkiller fasinumab.
“The development of novel pain medicines, such as fasinumab, can be one important step in combating the growing opioid epidemic,” said Dr. George D. Yancopoulos, Chief Scientific Officer, Regeneron and President, Regeneron Laboratories. “Fasinumab represents the culmination of more than 25 years of Regeneron scientific work in neurotrophic factors.”





Join or login to leave a comment
JOIN LOGIN