Medical imaging device company MolecuLight is a global leader in applying fluorescence imaging to detect and localize elevated bacterial burden in wounds. Their point-of-care devices allow clinicians to safely visualize the presence and location of high bacterial loads in real time so that clinicians can direct their wound care in the best possible way.
The Toronto-based company has recently announced positive results from an independent clinical trial which showed that their fluorescence imaging device MolecuLight i:X improved 12-week wound healing of diabetic foot ulcers by 204 percent.
This month, MolecuLight has secured financing from BDC Capital and iGan Ventures to support the company’s global expansion to meet the increasing market demand for their i:X and DX devices.
Xtalks learned more about MolecuLight and their i:X and DX devices through an email interview with MolecuLight’s CEO Anil Amlani. Read on to learn more!
What is MolecuLight’s mission?
Amlani: Wound care is a massive clinical problem worldwide and the specialty is plagued by a lack of real-time information on the state of bacterial burden in wounds. MolecuLight’s mission is to improve the state of wound care globally and help improve clinical outcomes. We are doing this by providing clinicians with the most useful information on their patients’ wounds in real time and at the point-of-care to inform their decision making. In essence, we are providing diagnostic imaging to wound care.
Please tell us a bit about MolecuLight’s history. When was the company founded?
Amlani: MolecuLight Inc. was founded in June 2012 by Dr. Ralph DaCosta, the Cancer Care Ontario Research Chair in Cancer Imaging and a Principal Investigator and Scientist at the Princess Margaret Cancer, University Health Network.
Dr. DaCosta applied his expertise in optical imaging to develop the MolecuLight i:X, the first portable point-of-care device to harness safe fluorescence light to visualize clinically significant bacterial burden in wounds. The idea was a merger of safe fluorescence light, sophisticated filtering systems to allow the device to only see clinically challenging bacterial burden, and miniaturization to create a device that was small, portable, highly reliable and easy to operate.
MolecuLight is now a global company, headquartered in Toronto, Canada with US headquarters in Pittsburgh, PA. We have a direct sales and clinical applications team in North America and 16 distributors in 18 countries to enable the sales and support for our products internationally.
The company has a significant body of peer-reviewed clinical evidence, including over 60 publications and more than 1,500 patients, showing the broad-based clinical utility and cost-savings that our MolecuLight devices are enabling — and we have a reimbursement pathway in the USA and South Korea for our MolecuLight procedure.
Most importantly, we have a large and growing user base of global clinicians that are using our devices to improve their wound care decision making — and ultimately improving outcomes for their patients worldwide.
How does MolecuLight stand out from other digital wound care/measurement companies?
Amlani: The cost of wound care is massive — it actually exceeds the cost of cancer. This includes diabetic foot ulcers — which plague one out of four persons with diabetes. Other types of wounds include pressure ulcers, venous leg ulcers, surgical site infections and burns.
Clinically significant bacteria is a massive problem because the presence of this bacteria precludes the healing of wounds and often renders many therapies ineffective.
The challenge is that other specialities have diagnostic imaging to assist clinicians. Orthopaedics have X-ray, cardiology has echocardiography, cancer has ultrasound, PET, CT and MRI. There is no real-time diagnostic imaging for wound care that allows clinicians to see the presence and location of elevated bacterial load. This is the clinical problem that MolecuLight is solving — and our imaging devices are making a global impact.
Many of our leading customers feel that MolecuLight is becoming a standard-of-care in point-of-care detection of elevated bacterial burden. There is no other company or product that has a FDA/CE/Health Canada-approved device and that has such a breadth of clinical evidence that supports the utility and outcomes that our devices are helping wound care clinicians to achieve.
In June this year, MolecuLight received an expansion to its US FDA 510(k) clearance for the MolecuLight i:X imaging device’s ability to detect the location of elevated bacterial loads in wounds and to identify areas of wounds containing more bacterial species of interest. What clinical value does this expanded clearance provide?
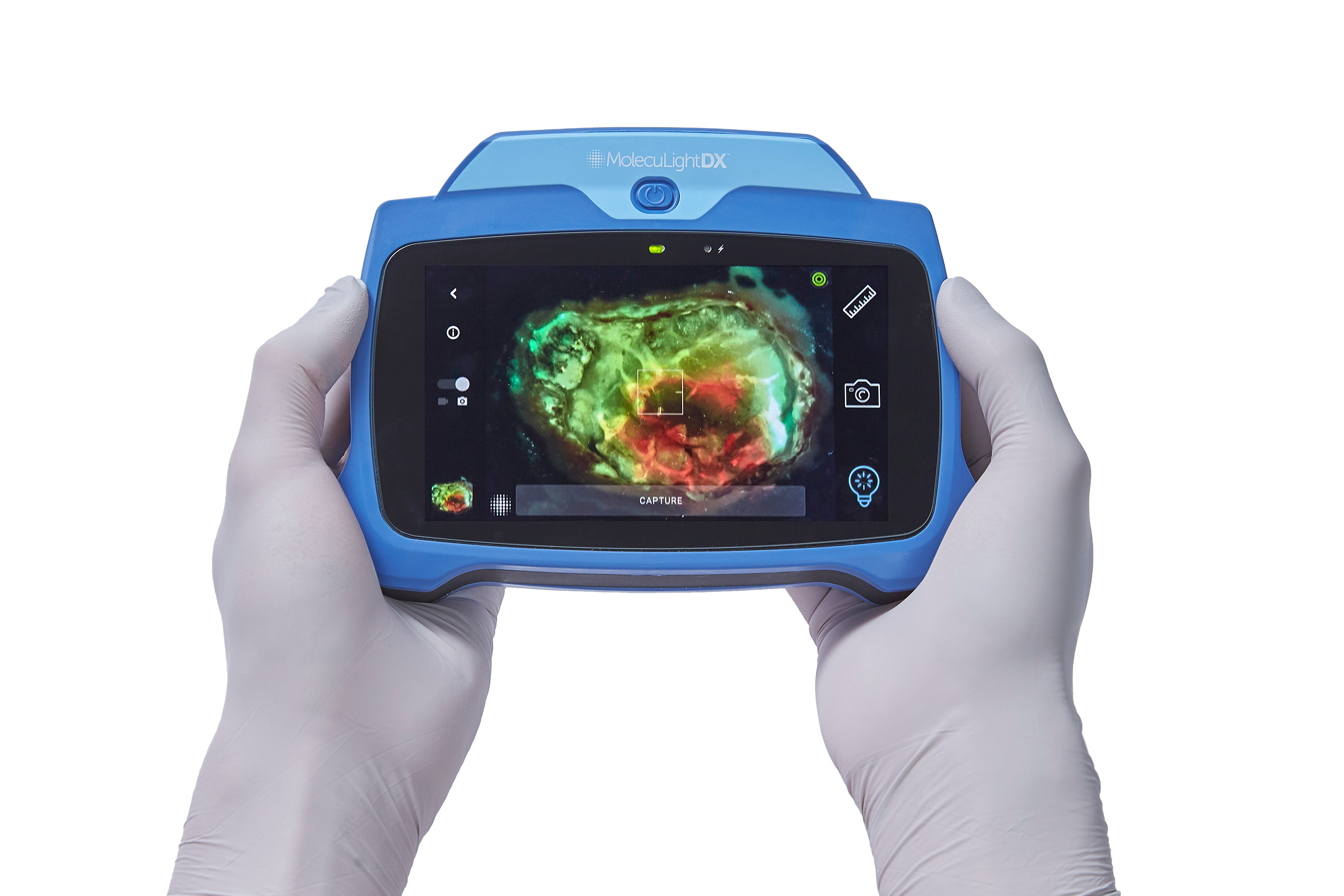
Amlani: We are thrilled with the FDA’s new clearance for MolecuLight’ ability to determine the location of elevated bacterial loads in wounds, in addition to the ability to identify regions with more bacterial species of interest. Clinicians worldwide are using the MolecuLight device to visualize regions with clinically significant bacterial loads and more species of concern. With point-of-care information on bacterial load and its locations through use of a MolecuLight device, clinicians can act immediately to tailor their cleaning, debridement, antimicrobial strategies and treatments accordingly.
This video (courtesy of Rose Raizman) illustrates the importance of visualizing the location of elevated bacterial load in a wound. In this scenario, the clinician is using MolecuLight i:X to inform their decision-making and target their wound hygiene to the areas of red fluorescence. Regions of red, indicating that the wound contains clinically significant (>104 CFU/g) levels of bacterial burden, are clearly visible on the patient’s diabetic foot ulcer.
MolecuLight was the first to receive FDA De Novo clearance for its MolecuLight i:X imaging platform and has subsequently received three additional FDA 510(k) clearances for the device, including this clearance. These clearances also apply to our latest device, the MolecuLight DX.
What are the next steps the company is working on for its MolecuLight i:X and DX devices in digital wound care?
Amlani: We have a really exciting product pipeline and significant R&D effort underway that is aimed at both the capturing of more information about the wounds on our devices, as well as the ability to better manage and leverage patient information to help clinicians understand and anticipate how wounds are healing.
This includes new imaging modalities to capture more imaging biomarkers, adding to our already comprehensive digital wound measurement toolkit, adding to our cloud-based software suite, and leveraging AI and machine learning to build predictive algorithms to help clinicians understand wound healing trajectories.











Join or login to leave a comment
JOIN LOGIN