In this episode, Ayesha discussed a new COVID-19 test technology that Innova Medical Group, world leader in at-home COVID-19 tests, has reached a licensing deal for with the University of Birmingham where the technology was developed. The new test is as sensitive and accurate as PCR tests and faster than lateral flow (rapid antigen) tests. Hear about the innovative new technology underlying the molecular test, and how the test is also being developed for the detection of other viruses.
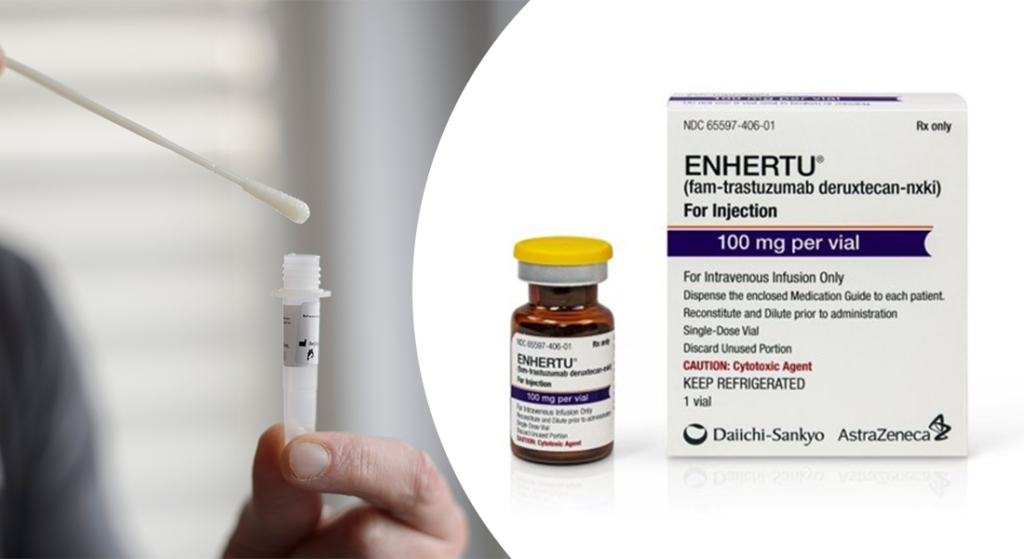
Ayesha also talked about the FDA approval of AstraZeneca and Daiichi Sankyo’s antibody-drug conjugate (ADC) Enhertu (trastuzumab-deruxtecan) for the treatment of patients with unresectable or metastatic HER2-low breast cancer. The approval makes Enhertu the first approved drug for this indication. Find out about the trial data that led to the approval and its effectiveness in HER2-low breast cancer, which is a newer subtype of the cancer.
Read the full articles here:
Innova Secures Licensing Rights for U of Birmingham’s New COVID-19 Test Technology RTF-EXPAR
AstraZeneca’s Enhertu Gets FDA Approved as First Therapy for HER2-Low Breast Cancer
The weekly podcast is available for streaming every Wednesday on Spotify, Apple Music and wherever you stream your podcasts.
Subscribe to the Xtalks Life Science Podcast to never miss a new episode.












Join or login to leave a comment
JOIN LOGIN