Update (September 14, 2018): Regeneron has now submitted a supplemental Biologics License Application (sBLA) to the FDA for Eylea to treat diabetic retinopathy. The pharma company hopes to hear back from the regulator by May 13, 2019.
Originally published on August 21, 2018:
The US Food and Drug Administration (FDA) had approved Regeneron’s supplemental Biologics License Application (sBLA) for a 12-week dosing regimen for Eylea (aflibercept) in the treatment of wet age-related macular degeneration. The medication was previously approved for the same indication with both four- and eight-week dosing intervals.
The approval comes after the FDA originally rejected the company’s sBLA, opting instead to issue a complete response letter to Regeneron. While the company said it could take about two months to complete discussions with the FDA and be granted approval, the turnaround time took just four days.
The approval was based on data from two Phase III studies – VIEW 1 and 2 – both of which were in their second year of treating patients with wet age-related macular degeneration with a 12-week dosing schedule for Eylea.
“We are pleased that the FDA has approved an updated label for Eylea,” said Dr. George D. Yancopoulos, President and Chief Scientific Officer of Regeneron. “Providing information to retinal physicians about the visual outcomes with a modified 12-week dosing schedule will help physicans make the most informed choices in treating patients suffering from wet age-related macular degeneration.”
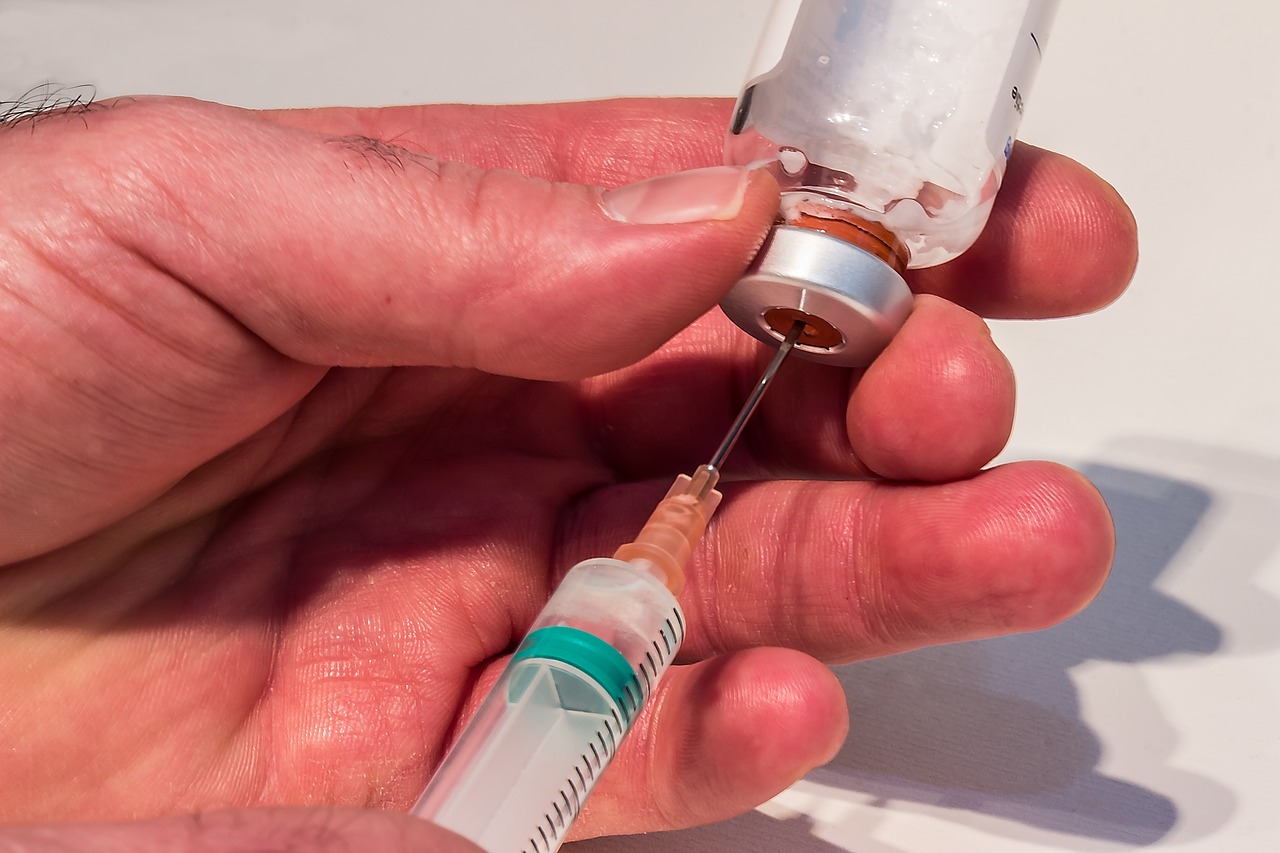
Eylea is an injectable drug containing a vascular endothelial growth factor (VEGF) inhibitor. Since the drug is delivered directly into the eye, it’s designed to restrict the growth of new blood vessels, known as angiogenesis, which reduces the amount of fluid build up in the eye that contributes to the progression of wet age-related macular degeneration.
Originally approved in 2011, Eylea brought in almost $6 billion in global sales for Regeneron in 2017. US sales of the treatment increased by 11 percent from 2016 to 2017 to reach $3.7 billion.
It’s estimated that up to 11 million people in the US suffer from some form of age-related macular degeneration, with vision loss occurring most often in those over the age of 60. While wet age-related macular degeneration is the less common of the two forms of the disease – accounting for just 10 percent of all cases – this type of macular degeneration makes up the vast majority of cases of eventual blindness.
In addition to its indication in macular degeneration, Regeneron is also chasing the diabetic retinopathy market for Eylea. The company released promising results from the Phase III trial in March of this year and plans to submit an sBLA to the FDA in late 2018.







Join or login to leave a comment
JOIN LOGIN