*The above authors contributed equally to this work
Therapeutic and diagnostic approaches involving the use of radiation and radioactive compounds have a long-standing history in the fields of science and medicine. Radiotherapy was first used in cancer treatments in 1896.1 Since then, the field of radiation has advanced to further understand how radioactive compounds interact with biological tissues and how they can be used in both diagnostic and therapeutic applications.
Radiopharmaceuticals are compounds used for medicinal purposes that contain radioactive isotopes (also known as radionuclides) and can be diagnostic or therapeutic in nature, or both.2 They represent a unique category of pharmaceuticals due to their radioactive properties. As such, there are specific guidelines and regulations that impact and direct the study and use of these compounds.
Radiopharmaceutical drug development has rapidly expanded over the last decade. Radiopharmaceuticals are widely used in the field of imaging for diagnosis, staging and follow‑up; in the realm of therapeutics, their use has increased, most notably, in the area of oncology.
In a recent webinar, experts from Medpace’s radiation oncology, imaging, regulatory and operational teams discussed the growing space of radiopharmaceutical development with respect to their biological use and application, regulatory frameworks that govern their evaluation in support of approvals, operational manufacturing considerations and associated imaging approaches.
Biological Mechanisms of Action of Radionuclides
According to Dr. Jess Guarnaschelli, Medical Director, Radiation Oncology, the radioactivity of radionuclides can be employed for both diagnostic and therapeutic medical uses. While external beam ionizing radiation involves radiation emitted in the form of electromagnetic waves or particles, radiopharmaceuticals use radionuclides to deliver localized radiation to specific targets.
The use of radiopharmaceuticals for therapy is based on the accumulation of an isotope within the target of interest. Then, radiation is delivered through the release of ions by alpha particles and beta particles. This induces cellular damage and death by either direct DNA strand breaks (single or double strand breaks), or indirect injury through the generation of reactive oxygen species (ie, free radicals) which can further damage molecules and DNA.3
There are many advantages of using radiopharmaceuticals in the treatment of diseases like cancer, some of which include:
- Effective and specific targeting of tumor cells
- Radionuclides with high linear energy transfer (LET) can effectively kill radioresistant cells (ie, hypoxic cells, or stem cells that often exhibit therapy resistance)
- Low whole body absorbed dose
- Various forms possible: free inorganic, conjugated to antibodies, small biomolecules
At the same time, there are also some disadvantages associated with radionuclides, some of which include non-uniform and uncontrolled distribution of radioactivity throughout the body, unwanted absorption by surrounding or distant tissues and organs, as well as changes in radiation dose rate due to decay.
Radionuclides have limited half-lives as they decay as energy is emitted. Administering a rapidly decaying product that has a very short half-life, which typically ranges in hours, presents challenges in its use. If the radiopharmaceutical agent becomes inert, it becomes ineffective without benefit to patients. The instability of radiolabeled compounds after administration is thus a major concern.
Current Clinical Uses of Therapeutic Radiopharmaceuticals
There are several different radionuclides that are used clinically. Among these are iodine-131, which has been used in the therapy of differentiated thyroid diseases for over half a century. Other clinically used radionuclides include strontium-89 and samarium-153 that can be used to treat and palliate painful skeletal metastasis.4
While radionuclide therapy can provide effective targeting, it can also have non-targeted effects. In evaluating the therapeutic ratio of radionuclide therapy, it is therefore important to understand and assess the net biological effect exerted on tumor and healthy cells, particularly during clinical trial scenarios.
One non-target effect of radionuclides is the impact of tumor heterogeneity on cell dose distribution in which the localized effect of the radionuclide only impacts a fraction of cells in tumor tissues. Tumors are typically heterogeneous as they consist of different clones or subpopulations of cells. Moreover, the spatial distribution of cells in a tumor can also have an impact; for example, cells at the periphery may receive a lethal dose of radiation while the center receives only a minimal dose. Layers of the tumor can also come into play, creating a dose gradient where some cells receive a lesser dose and become radioresistant as a result, which has implications for cancer recurrence. All of this quickly leads to a very complex scenario.
Targeting Mechanisms of Radiopharmaceuticals
Because benefits of radiotherapy could potentially be outweighed by damage to healthy tissue, proper targeting to area(s) of interest/tissue is critical.5 Radionuclide agents can be targeted to various locations within cells. It has been found that the cell membrane, in particular, is a preferred site of targeting. Tumor-specific antibodies or peptides can be used to facilitate targeting, as well as carrier molecules such as nanoparticles or liposomes that can deliver the radionuclide, along with other molecules, into the cell. These can enter through endocytosis, diffusion (if small enough), or membrane fusion. Radionuclides can be targeted to the nucleus through cyclic citrullinated peptide (CPP)-tagged antibodies or peptide-based analogs of DNA. Radionuclides can also be targeted to cell and nuclear membrane receptors.6
In order to design better clinical trial strategies to improve treatment efficacy and limit radiation side effects, Guarnaschelli said that “non-target effects, tumor heterogeneity, dose distribution and adaptive responses should be understood and taken into consideration when designing these clinical trials.” For this, there should be a significant focus on using dosimetry to quantitate assessment of tumor dose and account for radiation exposure of normal tissue. A well-trained and knowledgeable clinical study team will understand and address all of these concepts and technicalities, which is important in trial design and execution.
Dr. Sanje Goonasekera, a Senior Regulatory Affairs Associate at Medpace, informed that regulatory requirements specific to the geographies of interest must also be considered in the overall development plan. He reminded that the unique properties associated with radiopharmaceuticals will usually result in additional regulatory considerations and therefore a thorough understanding of the country-specific regulatory requirements will be key to avoid delays in study startup.
Regulatory Frameworks for Developing Radiopharmaceuticals
Although most major geographies have regulatory frameworks in place to provide guidance, a lack of harmonization across the geographies continues to be prevalent. A brief look at the United States (US) and European Union (EU) regulatory frameworks for radiopharmaceuticals can highlight how regulatory requirements can vary across regions and states. The applicable regulatory pathway for a given geography will be based on the type of product, its intended use, and associated risks.
Given these circumstances, Goonasekera said that it is important to “encourage sponsors to engage regulatory authorities early in the development cycle to gain alignment on the clinical development plan.”
US Regulatory Framework
There are several Agencies that have regulatory oversight of radiopharmaceuticals in the US. These include the Food and Drug Administration (FDA) and the Nuclear Regulatory Commission (NRC). While the NRC licenses the possession and use of radioactive material in the US, the FDA authorizes the use of radiopharmaceutical and diagnostics in investigational trials.7
Prior to commencing a clinical study for an investigational radiopharmaceutical, Sponsors are required to obtain Institutional Review Board (IRB) approval and file an Investigational New Drug (IND) application with the Agency. The specific division or office at the FDA that will have jurisdiction over a particular product will depend on the type of product and indication, “for example, if you are developing a diagnostic radiopharmaceutical, the Division of Medical Imaging and Radiation Medicine will have jurisdiction, while the relevant division at the Office of Oncologic Diseases will have oversight of a therapeutic radiopharmaceutical intended for the treatment of an oncologic disease” says Goonasekera.
Finally, pathways to market approval for radiopharmaceuticals in the US are no different than for any other standard pharmaceutical. In the past, Sponsors have sought approval via New Drug Applications (NDAs) and Biologics License Applications (BLAs). In addition to the 505(b)(1) pathway supporting an application containing full reports of safety and efficacy, successful examples of approval are available using the 505(b)(2) and the 505(j) pathways, via the Abbreviated New Drug Application (ANDA) .
EU Regulatory Framework
In the EU, radiopharmaceuticals are regulated as medicinal products and per the Clinical Trial Directive, approvals from the National Competent Authority and the Ethics Committee in the country of interest are required in order to initiate a clinical trial. In addition, although the overarching regulatory framework is based on the rules and regulations outlined in the EudraLex, country specific requirements may vary resulting in heterogeneity in study start-up requirements and timelines.8
Additionally, stakeholder agencies such as the European Association for Nuclear Medicine have released Good Radiopharmacy Practice guidelines. Although non-binding, these guidelines may be considered in specific member states.9
In the EU, all medicines must be authorized before they can be placed on the market. Applicants can seek marketing authorization for a radiopharmaceutical in a given country via the national procedure, or seek marketing authorization in more than one state via the mutual recognition or the decentralized procedures. Finally, applicants can seek marketing authorization via the centralized procedure, following review by the European Medicines Agency (EMA). It is worth to note that the centralized procedure is compulsory for some therapeutic indications, such as oncology.8
The New Clinical Trial Regulation, which came into force in 2014, will replace the Clinical Trial Directive, and is expected to become applicable towards the end of 2021.10
The new regulations will aim “to harmonize clinical trials within the EU, and changes will be seen mostly for diagnostic radiopharmaceuticals,” said Goonasekera. “Planning ahead for the changes will be critical as sponsors will have three years for the transition,” he added.
Some of the key changes associated with the new regulatory framework that may impact diagnostic radiopharmaceuticals are as follows:11
- Manufacturing and import authorization will no longer be needed for diagnostic radiopharmaceuticals prepared in accordance with the manufacturer’s instructions for use in study sites participating in the same clinical trial in the same member state.
- Good Manufacturing Practice (GMP) production in accordance with EudraLex Volume 4 will not be a requirement for diagnostic radiopharmaceuticals. However, these can still be imposed by national regulations.
- Simplified labeling requirements for primary packaging.
In addition to the high-level requirements for initiating a clinical trial and obtaining market approval, specific Chemistry Manufacturing and Controls (CMC) and non-clinical considerations can also apply when developing radiopharmaceuticals. For example, because of the unique biophysical properties associated with radioisotopes used with positron emission tomography (PET), these drugs are outlined separately in the Code of Federal Regulations and are considered a special category with specific GMP requirements. In the EU, EudraLex Volume 4, Annex 3 lists GMP guidelines for radiopharmaceuticals that also includes PET drugs.12
Both US and EU have recently released guidelines covering various aspects of non‑clinical development, and while they complement what is already available on non-clinical development, they also align for the most part. For example, both FDA and EMA appear to agree that it may be sufficient to conduct toxicology studies in one relevant species, and that given the well-known potential for these agents to cause DNA damage, reproductive and carcinogenicity studies may be waived.13,14,15 Even with these guidelines, it is advised to obtain agreement by regulators on the completeness of a non-clinical package.
When it comes to Good Laboratory Practice (GLP) requirements, the US and EU may not see eye‑to-eye. Given the challenges of conducting some of these studies and based on existing literature, FDA more recently has shown some flexibility in accepting non-GLP toxicology studies.16 “However, to our understanding, this is not the case as of yet in Europe,” says Goonasekera.
Dual-Functioning Radiopharmaceuticals: Theranostics
A growing area of interest in radiopharmaceuticals is the concept of a theranostic, which is the combination of a targeted therapeutic with a targeted diagnostic, also referred to as the “see‑and‑treat” approach.17 A theranostic could be a single isotope with multiple emitting capacities such as in the case of iobenguane iodine-131 (Azedra®), or it could be a combination of different isotopes each with unique emitting capacities, such as in the case of lutetium-177 and galium-68 used together to diagnose and treat neuroendocrine tumors. A theranostic pair could be either co‑developed (parallel model) or developed sequentially (in-series model) and each of these pathways will have specific regulatory considerations to take into account.18
Operational Considerations in Radiopharmaceutical Clinical Trials
Study start-up for radiopharmaceutical trials is highly complex, with many additional factors to consider. A main rate-limiting step is often regulatory requirements: the significant heterogeneity in regulatory frameworks across regions, coupled with specificities regarding site radioactivity licenses and locally required additional authorizations, means these factors need to be thoroughly evaluated when planning submissions. In addition to this, there are multiple start-up tasks specific to radiopharmaceutical trials that must also be handled in parallel: manufacturing qualification, imaging qualification, provision of highly specialist supplies and practice investigational product shipments are but a few of the tasks that can prevent or delay site start-up.
Careful research to understand the pertinent regulations, pre-planning of all activities and proactive communication between all parties is therefore key in ensuring an optimal study start-up. As stated by Stephanie Millin, Clinical Trial Manager at Medpace, “these factors are largely predictable, so with the right knowledge and the right strategy, they can be managed successfully.”
Supply Chain Considerations
There are various factors that dictate supply chain manufacturing of radiopharmaceuticals. One of the most important factors among these is the half-life of the product. A product with a relatively long half-life, and/or that has specific manufacturing requirements, is usually manufactured centrally. On the other hand, radionuclides with short half-lives are produced locally in order to facilitate timely production, delivery, and use. Each type is associated with a unique set of challenges in management, and the logistics involved in the manufacturing, distribution, and administration of the investigational product are, therefore, of great importance.
Some products, generally those with a short half-life, can be supplied as a non-radioactive or ‘cold’ kit, which can be stored at the trial site or local radio pharmacy and reconstituted and labeled with a radionuclide on demand. As manufacturing will occur at the site, it is important to ensure sufficient processes are in place to guarantee the quality of the final product.
Products manufactured centrally generally have a shelf life of a few days, but are calibrated to be administered at a specific date and time which cannot be deviated from. Robust supply chains to guarantee on-time delivery, in addition to site expertise to manage patient dosing to the minute, are critical.
Study Logistics and Patient Engagement
Radiopharmaceutical studies require collaboration from several departments including nuclear medicine and radiopharmacy, oncology, radiology and more. Procedure schedules can also be highly demanding, often including a high number of time-critical procedures and imaging timepoints. Administration and preparation data for the investigational product, imaging and blood and urine analyses for radioactivity at specified timepoints are among some of the additional elements and data that must be collected in a radiopharmaceutical trial to fully achieve primary endpoints. Communication and proactive planning are key to success and it is therefore paramount to actively engage all involved personnel from the outset.
Patient management and engagement is also an important factor to consider. Given the high demands placed on the patient coupled with a potential impact on their home life, such as their proximity to family and pets, it is important to provide the needed support and education so that the study is as easy and accessible to them as possible.
Each part of a radiopharmaceutical clinical trial involves a lot of tasks in parallel, many of which are dependent on others, creating significant logistical complexity. Millin adds that for each activity, “it’s a lot to manage, with time being critical and with a lot of specialist staff and equipment.” To manage this efficiently she explains that, “it’s important to clearly delineate roles and responsibilities and communication pathways upfront to ensure all planning is thorough.”
Imaging Requirements and Considerations
Imaging is a significant component in the use and application of radiopharmaceuticals. It is therefore a critical aspect to consider in radiopharmaceutical clinical trials. The most important aspect before scanning the first patient included in a specific radiopharmaceutical trial, is scanner calibration. It is required to obtain a comparable quantification in multicenter settings. For diagnostic radiopharmaceuticals, PET imaging calibration aims to achieve standardized uptake value (SUV) harmonization. For therapeutic pharmaceuticals, planar or single-photon emission computed tomography (SPECT) imaging calibration aims to standardize the acquisition parameters. This calibration is essential for clinical trials requiring dosimetry and/or SUV calculation.
Sites must strictly follow imaging protocols as no deviation is permitted for further data analysis. This is even more important as a patient cannot be re-scanned.
An example of diagnostic radiopharmaceuticals is the use of 68Ga-DOTATOCDOTATOC that targets somatostatin receptors that are over-expressed in patient with metastatic small cell lung carcinoma.
An example of the association of diagnostic and therapeutic radiopharmaceuticals, also called theranostic, is the use of both 68Ga-prostate specific membrane antigen (PSMA) and 177Lu-PSMA. Prostate specific membrane antigen is an antigen overexpressed by prostate cancer cells. The 177Lu-PSMA images show uptake within lesions corresponding to those seen on the 68Ga-PSMA PET image.
Another example of a therapy agent is the use of 177Lu-DOTA-TATE (targets somatostatin receptors). After several cycles of therapy, this internal radiotherapy has been shown to dramatically reduce the size of the primary lesion.
In regards to the use of planar (2D) vs SPECT/CT (3D) images, some of the advantages and disadvantages include: planar allows the whole body to be scanned in about 30 minutes, however, for dosimetry calculation, there is a need for a transmission scan and the total volume of patient’s urine to be collected. In SPECT/CT, the whole body cannot be scanned as it will take too much time. One field of view (FOV), covering about 40 cm of the patient’s height, takes 15 minutes to be scanned. The CT scan from the SPECT/CT scanner already provides a transmission scan and the 3D acquisition allows for more accurate dosimetry calculation than 2D planar. Radiation dosimetry is used to estimate the absorbed dose in Gy/GBq of a radioactive compound in critical organs and/or tumors. It helps to define an efficient treatment dose without damaging critical organs. It can also help to predict a treatment dose based on a diagnostic agent using the same pharmaceutical compound.
The following is an example of time activity curves for a few different organs (Figure 1). The activity in MBq/g is expressed as a function of time after injection (Mins p.i):
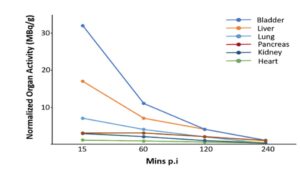
Below (Figure 2) is an example of the absorbed dose for a few organs (Gy/GBq). The kidney is the most critical organ in this example.
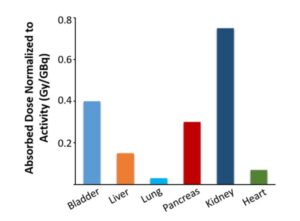
To conclude, imaging is an essential component of radiopharmaceutical-based therapies. It is therefore important to ensure that scanners are calibrated and that sites follow appropriate and correct imaging protocols to ensure consistency and accuracy, which is important for image quality and dosimetry calculations. SPECT/CT should be considered instead of planar if dosimetry has to be calculated, and imaging time points have to be defined with the dosimetrist before protocol finalization. Also, the FOV has to include source organs for dosimetry. Finally, dosimetry calculation is a powerful tool and can help physicians in their clinical decisions for treatment dose.
Radiopharmaceuticals in Diagnostics and Treatment: Considerations in the Evolving Landscape
In the era of targeted therapies, radiopharmaceuticals are highly attractive therapeutic delivery systems as they can target tumor cells to allow for their simultaneous imaging and radio-treatment. Medpace is driven and well-equipped to assist in the planning and oversight of trials in the radiopharmaceutical arena. There are a number of biological, clinical, regulatory, operational and imaging considerations that must be factored in when designing a radiopharmaceutical clinical trial. These elements were discussed in Medpace’s informative webinar on the evolving landscape of radiopharmaceuticals. Obtaining the necessary knowledge and information, along with careful and early planning, is essential in the design of a robust trial that mitigates risks and optimally achieves target endpoints.
References
1. Holsti LR. Development of Clinical Radiotherapy Since 1896. Acta Oncologica 1995: 34(8):995-1003.
2. Munjal A, Gupta N. Radiopharmaceuticals. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2020.
3. Hall EJ, Amato JG. Radiobiology for the Radiologist. Philadelphia: Wolters Kluwer Health/Lippincott Williams & Wilkins, 2012.
4. International Atomic Energy Agency https://www.iaea.org/resources/rpop/health-professionals/nuclear-medicine/therapeutic-nuclear-medicine/specific-therapies. Accessed 14 May 2020.
5. Hubenak JR, Zhang Q, Branch CD, Kronowitz SJ. Mechanisms of injury to normal tissue after radiotherapy: a review. Plast Reconstr Surg. 2014;133(1):49e‐56e.
6. Kumar C, Shetake N, Desai S et al., Relevance of radiobiological concepts in radionuclide therapy of cancer, Int J Radiat Biol. 2016:92(4):173-186.
7. Petri N. NRC and FDA Regulations Affecting Nuclear Pharmacy Practice. J Pharm Pract. 1989; Vol II:306-313.
8. Sharma S et al. Intricacies in the Approval of Radiopharmaceuticals – Regulatory Perspectives and the Way Forward. Current Science, 2019; 116(1):47–55.
9. Elsinga P, Todde S, Penuelas I, et al. Guidance on current good radiopharmacy practice (cGRPP) for the small-scale preparation of radiopharmaceuticals. Eur J Nucl Med Mol Imaging. 2010;37(5):1049‐1062.
10. Billiones R. “2020: Will the EU clinical trials regulation be implemented this year?” https://www.linkedin.com/pulse/2020-eu-clinical-trials-regulation-implemented-year-raquel-billiones/. Accessed 7 April 2020.
11. Penuelas I et al. The New Regulation on Clinical Trials in Relation to Radiopharmaceuticals: When and How Will it be Implemented? EJNMMI Radiopharm Chem, 2019; 11;4(1):2.
12. EudraLex, The Rules Governing Medicinal Products in the European Union, Volume 4, EU Guidelines to Good Manufacturing Practices, Annex 3, Manufacture of Radiopharmaceuticals.
13. European Medicines Agency. Committee for Medicinal Products for Human Use. Guideline on the Non-Clinical Requirements for Radiopharmaceuticals. Draft Guideline. October 2018.
14. US Department of Health and Human Services. Food and Drug Administration, Center for Drug Evaluation and Research. Microdose Radiopharmaceutical Diagnostic Drugs: Nonclinical Study Recommendations. Guidance for Industry. August 2018.
15. US Department of Health and Human Services. Food and Drug Administration, Center for Drug Evaluation and Research. Oncology Therapeutics Radiopharmaceuticals: Nonclinical Studies and Labeling Recommendations. Guidance for Industry. August 2019.
16. Schwartz S and Decristoforo C. US and EU Radiopharmaceutical Diagnostic and Therapeutic Nonclinical Study Requirements for Clinical Trials Authorizations and Marketing Authorizations. EJNMMI Radiopharm Chem, 2019; 4:10.
17. Yordanova A et al. Theranostics in Nuclear Medicine Practice. Onco Targets and Therapy, 2017; 10:4821–4828.
18. Theranostics: Regulatory Considerations for Product Development. Society of Nuclear Medicine and Molecular Imaging. Annual Meeting, Anaheim California
This article was created in collaboration with the sponsoring company and the Xtalks editorial team.





Join or login to leave a comment
JOIN LOGIN