The limelight is now on Vtama (tapinarof) cream for the treatment of plaque psoriasis. This cream by Dermavant is the first steroid-free topical medication in its class.
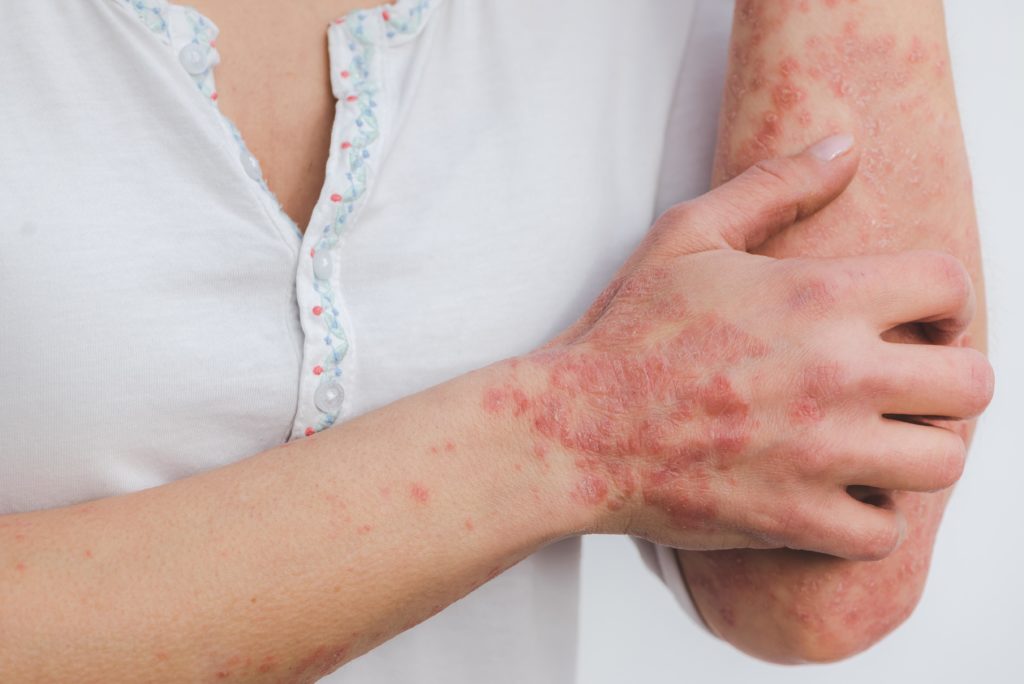
Psoriasis is a well-recognized skin disease. It has been estimated that around eight million people in the US and 125 million people in the world have been affected by psoriasis. This immune-mediated inflammatory disease has been researched thoroughly. The precise cause of psoriasis is unknown, but risk factors include genetics and a family history of the condition, heavy alcohol consumption, stress, smoking and dry or cold weather. There are five distinct types of psoriasis, namely plaque, guttate, pustular, inverse and erythrodermic psoriasis.
Psoriasis is considered a life-long ailment with no permanent cure. The disease usually develops around 15 to 25 years of age and can recur at any point of time in the affected individual’s life. Individuals with psoriasis are at a higher risk of developing other medical conditions, such as psoriatic arthritis, Crohn’s disease, inflammatory bowel disease, hypertension, diabetes and psychological/psychiatric disorders.
Current treatment methods of psoriasis include topical therapy (e.g., corticosteroids, vitamin D3 analogs, calcineurin inhibitors), light therapy (ultraviolet B [UVB] phototherapy, excimer laser) and oral or injected medications (e.g., immunomodulatory agents [biologics], oral retinoids, methotrexate). Topical applications have been studied profoundly and were found to have limitations, like in their duration and extent of use.
The spotlight for plaque psoriasis treatment is now on Vtama (tapinarof) cream (one percent), Dermavant’s discovery that has become the first steroid-free topical medication in its class approved by the US Food and Drug Administration (FDA).
The Chemical Composition of Vtama
The active ingredient in Vtama topical cream is tapinarof, and there are several other inactive ingredients such as benzoic acid, butylated hydroxytoluene, citric acid monohydrate, diethylene glycol monoethyl ether, edetate disodium and more. Tapinarof was discovered as an outcome of investigations into the secondary metabolites of Photorhabdus luminescens, a bioluminescent, Gram-negative bacteria. There is 10 mg of tapinarof per gram of Vtama cream.
XTALKS WEBINAR: All Means All: The Road to Inclusivity in Clinical Trials
Live and On-Demand: Tuesday, July 12, 2022, at 11am EDT (4pm BST/UK)
Register for this free webinar to learn what the FDA’s new draft guidance means for diverse and inclusive trials. The featured speakers will discuss its shortcomings and what the industry still needs to address to make clinical trials more inclusive for all.
Immunological Response to Tapinarof
The efficacy of tapinarof has been proved by its binding efficiency with the aryl hydrocarbon receptor (AhR). The AhR is a ligand-activated transcription factor, and it is considered to play a vital role in skin homeostasis. The naturally-derived small molecule tapinarof has been found to bind to the AhR and activate it, which contributes to the downregulation of the immune-related factors such as proinflammatory T helper type 17 (Th17) and Th22 cytokines (interleukin-17 [IL-17A] and IL-22). Tapinarof treatment in a mouse model of psoriasis resulted in the downregulation of inflammatory cytokine expression in skin tissue, which involved IL17A, IL17F, IL19, IL22, IL23A and IL1B gene expression.
Clinical Trials of Vtama
Dermavant assessed the efficiency of Vtama for plaque psoriasis by conducting two multicenter, randomized, double-blind, vehicle-controlled trials named PSOARING 1 (NCT03956355) and PSOARING 2 (NCT03983980) which were each 12 weeks long. In addition, a long-term extension (LTE) study named PSOARING 3 (NCT04053387) was also done, which involved 40 weeks of open-label treatment with Vtama cream followed by a four-week follow up.
At week 12 of PSOARING 1 and PSOARING 2, Vtama proved to establish a score of “clear” or “almost clear” in a statistically significant number of patients when compared to vehicle. The Psoriasis Area and Severity Index (PASI) score with Vtama cream had more than 75 percent improvement from baseline at week 12.
More than 40 percent of the PSOARING 3 participants had complete disease clearance at least once during the study period. For the participants that saw clear skin during PSOARING 1 or PSOARING 2 and enrolled in PSOARING 3, Vtama cream had a remittive effect maintaining clear or almost clear skin with a median time to first worsening of around four months after cessation of treatment.
“At Dermavant, we are committed to advancing novel, patient-focused innovation in immuno-dermatology. As such, we are proud to have developed a topical treatment in Vtama cream that provides not only efficacy over 52 weeks but can also be used on all body areas, including on sensitive locations, such as face, skin folds, neck, genitalia, anal crux, inflammatory areas and axillae. In addition, an approximately four month off-treatment remittive effect (median time to first worsening), leads us to believe that Vtama cream has the potential to become the preferred topical option for this chronically underserved patient population and among the physicians who treat them,” said Todd Zavodnick, Chief Executive Officer of Dermavant, in the company’s press release.
Dermavant’s Future Targets
Dermavant has started a Phase IIA clinical trial (NCT04103060) to evaluate the safety and tolerability of cerdulatinib (DMVT-502) in adults with vitiligo. The company is currently recruiting participants for a Phase III study (NCT05014568) to evaluate the safety and efficacy of topical tapinarof cream (one percent) in pediatric and adult subjects with atopic dermatitis. Oxybutynin/pilocarpine (DMVT-504) is an oral combination of an immediate-release muscarinic antagonist with a delayed-release muscarinic agonist that is currently under clinical trials for primary focal hyperhidrosis.
Dermavant has two drugs in preclinical development: DMVT-503, an investigational drug for acne vulgaris and lotamilast (DMVT-501), a highly selective topical PDE4 inhibitor under investigation for the treatment of atopic dermatitis.











Join or login to leave a comment
JOIN LOGIN