Kite Pharma, a biopharma company specializing in cell therapy research that was acquired by Gilead Sciences in 2017, announced on Friday that it received approval from the US FDA for Tecartus, a cell-based gene and immune therapy, for the treatment of adults with refractory or relapsed Mantle Cell Lymphoma (MCL).
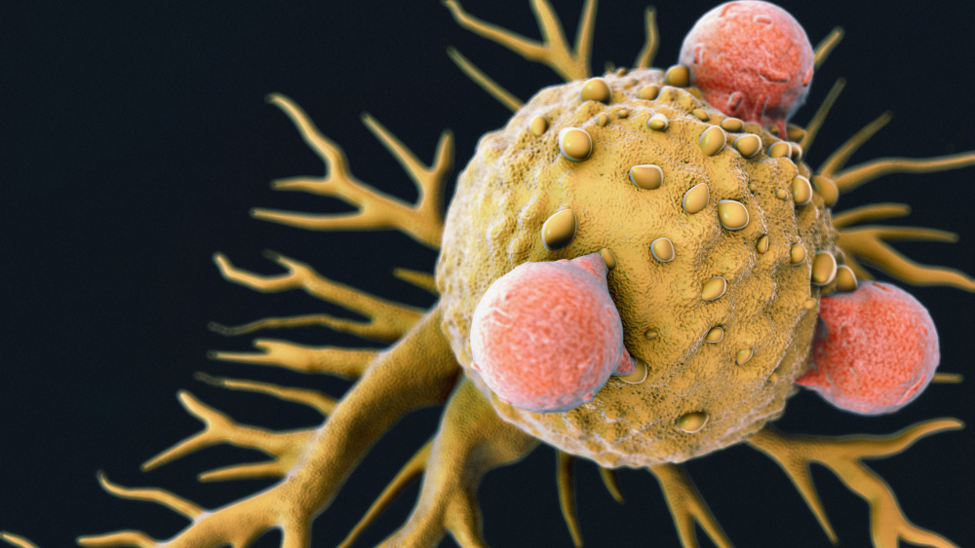
Tecartus is an anti-CD19 chimeric antigen receptor (CAR) T cell therapy that targets malignant CD19-expressing B cells in MCL. With the FDA approval, it has become the first cell-based CAR-T treatment for MCL, and the second approved CAR-T therapy from Gilead – with the first being Yescarta which is used in the treatment of diffuse large B-cell lymphoma (DLBCL).
“Tremendous progress has been made in the discovery of new therapies for debilitating diseases that are difficult to treat. This approval is yet another example of customized treatments that use a patient’s own immune system to help fight cancer, while using a scientific advance in this promising new area of medicine,” said Peter Marks, director of the FDA’s Center for Biologics Evaluation and Research (CBER). “We’re seeing continued advances in the field of gene therapy and remain committed to supporting innovation in this promising new area of medicine.”
MCL is a rare form of B-cell non-Hodgkin’s lymphoma (NHL) that develops along the outer edge of a lymph node follicle called the ‘mantle zone.’ B cells that reside in the mantle zone area become malignant, leading to the cancer in the lymph nodes, which can then rapidly spread to other parts of the body.
MCL occurs more commonly in men compared to women, and usually affects middle-aged or older adults, who are typically in their 60s.
Related: Venetoclax Combination Treatment Improves Overall Survival in Acute Myeloid Leukemia
Each Tecartus dose is designed to be tailor-made as a patient’s own immune system is harnessed to help fight the lymphoma. The immune cell therapy involves collecting a patient’s T cells and genetically modifying them by inserting a new gene that assists in targeting and killing lymphoma cells. These modified T cells are then injected back into the patient.
Tecartus uses technology that includes T cell selection and lymphocyte enrichment, which is “necessary in B cell malignancies such as mantle cell in which there are circulating lymphoblasts in the peripheral blood,” said Ken Takeshita, Kite’s global head of clinical development.
“The manufacturing method was tailored to this particular type of lymphoma,” he said, adding that after an MCL patient has failed on chemo and BTK inhibitors, “the prognosis is very poor, about a year or less.”
A multicenter Phase II clinical trial involving 60 adult patients with refractory or relapsed MCL demonstrated the safety and efficacy of Tecartus. The patients were followed for at least six months after their first objective disease response. Treatment with Tecartus led to a complete remission rate of 67 percent, most of whom were heavily pretreated, among the patients, and led to a 93 percent response rate. More than half of the patients continued to benefit from the treatment at a median of 12.3 months. Moreover, 43 percent of those that were monitored for at least two years post-treatment were still in remission without additional therapy.
To further evaluate the long-term safety of Tecartus, the FDA is requiring that the company conduct a post-marketing observational study involving patients treated with Tecartus.
The drug’s boxed label has warnings for the potential induction of cytokine release syndrome (CRS), a systemic immune response to the activation and expansion of CAR-T cells that causes high fever and flu-like symptoms. There is also a warning for neurologic toxicities, according to the FDA’s press release. Both CRS and neurologic toxicities can be fatal or life-threatening. Apart from these potential severe effects, Tecartus’ most common side effects include serious infections, low blood cell counts and weakening of the immune system.
“Because of the risk of CRS and neurological toxicities, Tecartus is being approved with a risk evaluation and mitigation strategy (REMS), which includes elements to assure safe use (ETASU). The risk mitigation measures for Tecartus are identical to those of the current REMS Program for another CAR-T therapy, Yescarta,” said the FDA.
The press release highlights that Tecartus was approved under the Accelerated Approval pathway and was granted Priority Review and Breakthrough Therapy designations. Tecartus also received Orphan Drug designation, which provides incentives to assist and encourage the development of drugs for rare diseases under FDA guidance.



Join or login to leave a comment
JOIN LOGIN