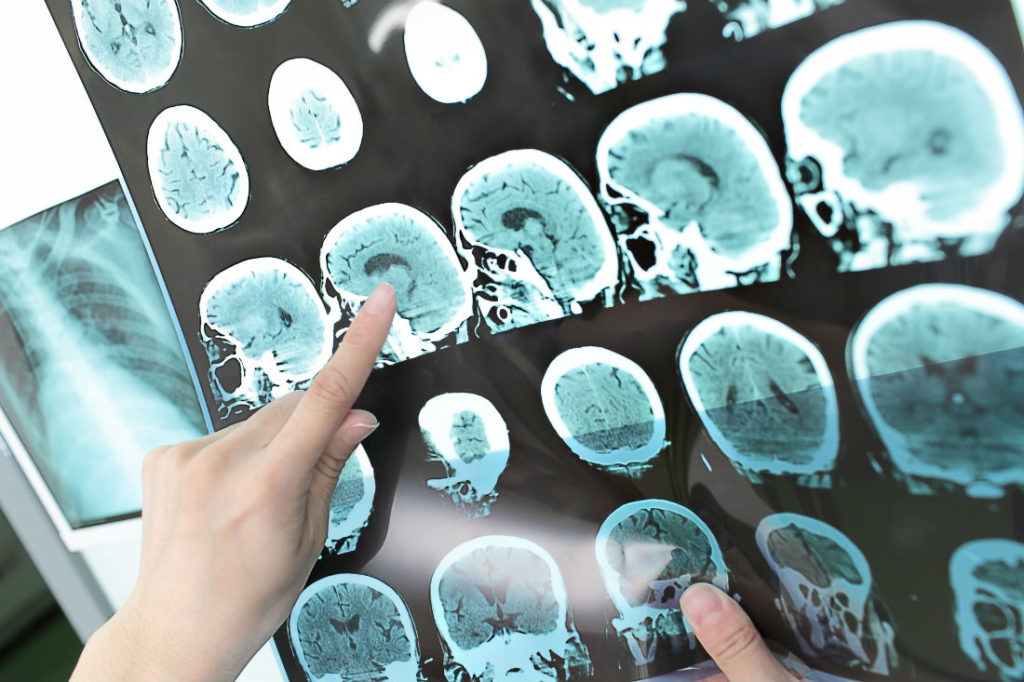
The FDA has just approved MaxQ AI’s Accipio Ix intracranial hemorrhage detection software which could help doctors diagnose patients with a brain bleed faster. The software uses artificial intelligence (AI) to analyze non-contrast head computed tomography (CT) scans and flag those that show signs of intracranial hemorrhage and should be prioritized by acute care physicians.
“We are very pleased to receive FDA 510(k) clearance for Accipio Ix, which we believe will be a transformative solution for medicine,” said Gene Saragnese, Chairman and CEO of MaxQ AI. “MaxQ’s Accipio application, of which Accipio Ix is the first part of the ecosystem, provides physicians with actionable intelligence, improving their future ability to make a timely, accurate and more confident diagnosis of a brain bleed.”
According to MaxQ AI, up to 30 percent of diagnoses based on medical imaging are incorrect, largely due to human error. To address this, AI is increasingly being used as a powerful tool to aid clinical decision-making to both improve patient outcomes and lower healthcare costs.
“Accipio Ix’s ability to greatly increase suspected bleed detection, improve escalation and time to review is a game-changer. Further, our partnerships with top CT and PACS companies including GE Healthcare, Samsung Neurologica and other organizations prior announced, make deployment and adoption of Accipio straightforward,” said Saragnese. “The potential impact is massive – only one patient diverted from acute stroke to wellness in the 13,000 US/EU hospitals represent $2 billion savings in a single year, and a lifetime of difference to the patient and their families.”
Using AI technology, the application can accurately detect signs of intracranial hemorrhage to help physicians prioritize patients that should be treated first. The Accipio Ix also has the benefit of keeping CT images in their original order to improve organization and it does not store any of the patients’ protected health information.
The clinical diagnostics intelligence platform company already received CE Mark approval in Europe earlier 2018. MaxQ AI plans to demonstrate the Accipio Ix system at the Radiological Society of North America (RSNA) 2018 Annual Meeting in Chicago.










Join or login to leave a comment
JOIN LOGIN