World Malaria Day 2026, which lands on April 25 every year, raises awareness about one of the world’s oldest and deadliest infectious diseases: malaria.
Despite decades of progress, the disease continues to exact a heavy toll, particularly in low- and middle-income countries, highlighting both how far we’ve come and how much work remains.
Why Does Malaria Persist Despite Being Preventable and Treatable?
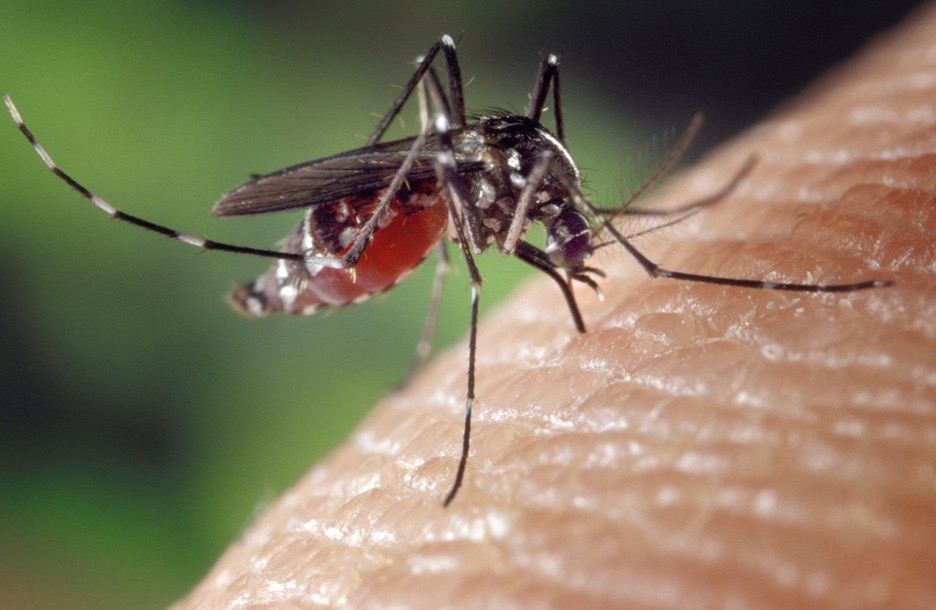
Malaria is caused by Plasmodium parasites transmitted through the bites of infected mosquitoes. While it is both preventable and treatable, it still disproportionately affects children under five and pregnant women, especially in sub-Saharan Africa.
According to the World Health Organization (WHO), hundreds of millions of cases occur globally each year, with the majority concentrated in a handful of high-burden countries. The persistence of malaria is not due to a lack of tools but rather gaps in access, infrastructure and sustained funding.
The infection threatens 3.3 billion people across 106 countries and causes over 600,000 deaths annually, mostly among African children. The day highlights the need for sustained investment, political commitment and innovative vector control to achieve a malaria-free world.
Once largely confined to tropical regions, malaria is now appearing in new geographies and re-emerging in areas where it was previously controlled, driven by shifting climates, expanding mosquito habitats and the rise of drug-resistant strains.
World Malaria Day 2026 with the WHO and Partners: Driven to End Malaria
On World Malaria Day 2026, the WHO has partnered with different organizations to launch the campaign: Driven to End Malaria: Now We Can. Now We Must.
The global health agency said the campaign “is a rallying cry to grasp the moment — to protect lives now and fund a malaria-free future.”
And it’s not all doom and gloom. The WHO says science is advancing faster than ever and that “for the first time, ending malaria in our lifetime is a real possibility.”
The agency is hopeful about new vaccines, treatments, improved malaria control tools and pioneering technologies, including genetic modification of mosquitoes and long-acting injectables, that are in development.
According to the WHO, 25 countries are rolling out malaria vaccines to protect 10 million children a year. Additionally, next-generation mosquito nets now make up 84% of all new nets distributed, according to the agency.
The WHO highlights that global malaria control has achieved major wins over the past two decades, with millions of cases and deaths averted thanks to expanded access to bed nets, rapid diagnostics and effective treatments.
Specifically, between 2000 and 2024, the number of malaria-endemic countries fell sharply, dropping from 108 to 80. Over the same period, 2.3 billion cases and 14 million deaths have been averted. To date, 47 countries have been certified malaria-free, while 37 countries reported fewer than 1,000 cases in 2024.
Success is possible, even in tough areas. The WHO cites the example of the Greater Mekong Subregion, which comprises Cambodia, China (Yunnan Province and Guangxi Zhuang Autonomous Region), Laos, Myanmar, Thailand and Vietnam, as an example where elimination is achievable, with cases falling by nearly 90% despite long-standing drug resistance.
“Nationally-led programs are driving change. The possibility has never been greater,” the WHO says.
Efforts to combat malaria increasingly rely on better data and coordination. Surveillance systems, real-world evidence and digital health tools are helping governments and global health organizations target interventions more effectively.
Partnerships led by organizations like the Roll Back Malaria (RBM) Partnership are aligning stakeholders, from governments to NGOs to the private sector, around shared goals of elimination. The WHO is a key partner and founding member of the RBM Partnership to End Malaria, which was established in 1998.
At its core, malaria is a disease of inequality and is linked to poverty and development. Communities most affected often face limited access to healthcare, diagnostics and preventive measures. Conflicts, displacement and fragile health systems further complicate the delivery of care.
World Malaria Day 2026 is about awareness and, importantly, a call to action to close these gaps. Expanding access to existing tools, investing in next-generation innovations and strengthening health systems will be essential to achieving long-term control and eventual eradication.
World Malaria Day 2026: What Is Driving Innovation in Malaria Treatment in 2026?
The malaria treatment landscape is undergoing one of its most significant shifts in decades, driven by rising concerns over drug resistance, particularly to artemisinin-based combination therapies (ACTs), which remain the global standard of care. In response, both industry and global health partners are accelerating the development of next-generation therapies, vaccines and long-acting prevention tools.
Artemisinin is a fast-acting antimalarial compound originally derived from the sweet wormwood plant (Artemisia annua), and it forms the backbone of current malaria treatment. It is typically used in combination therapies to rapidly reduce parasite levels in the blood while a partner drug clears remaining infection and helps prevent resistance.
While artemisinin itself wasn’t “approved” as a standalone drug in most major markets, instead, its derivatives became widely adopted through artemisinin-based combination therapies (ACTs).
The WHO began recommending ACTs as first-line treatment for uncomplicated malaria in 2001, which effectively marked their global clinical adoption. Individual artemisinin derivatives had already been in use in China since the late 1970s-1980s, but global-scale uptake came after WHO endorsement.
What Are the First Malaria Vaccines and How Effective Are They?
The rollout of the world’s first malaria vaccines, RTS,S/AS01 (Mosquirix) in 2021 and R21/Matrix-M in 2023, marks a historic shift in prevention efforts.
The WHO-recommended vaccines are being deployed in multiple African countries. Together, they represent the first generation of malaria vaccines to enter routine immunization programs at scale.
The vaccines significantly reduce severe disease and deaths in young children. According to the CDC, they reduce the risk of uncomplicated malaria by ~40%, severe malaria by ~30% and all-cause mortality by 13%.
Expanded rollouts are expected to play a growing role in reducing seasonal peaks of transmission.
The vaccines, combined with long-standing interventions such as insecticide-treated bed nets, rapid diagnostic testing and ACTs, are helping to reduce severe disease and deaths. Still, challenges like parasite resistance and climate-driven shifts in mosquito habitats threaten to erode these gains.
Next-Generation Antimalarial Drugs
One of the most closely watched recent developments in the malaria space is the emergence of new drug classes designed to overcome resistance to current ACTs.
Novartis is developing ganaplacide-lumefantrine (GanLum) in collaboration with the Medicines for Malaria Venture. In the latest results from a Phase III trial, the regimen has demonstrated cure rates above 97%. If all goes well, it will be the first major new antimalarial combination in over two decades.
GanLum is a combination of two compounds: the novel agent ganaplacide and a new once-daily formulation of existing antimalarial lumefantrine, a longer-acting treatment.
Ganaplacide belongs to a class of compounds called imidazolopiperazines, which Novartis said were first identified in their labs in San Diego, California, as potential antimalarials after a screen of 2.3 million molecules. The compound is thought to work by disrupting the parasite’s internal protein transport systems.
At the same time, researchers are exploring other new chemical classes beyond artemisinin derivatives, aiming to build multi-drug regimens that reduce the likelihood of resistance development. These efforts are critical as partial resistance to artemisinin has now been confirmed or suspected in multiple African countries, raising concerns about long-term treatment efficacy.
Next-Gen Malaria Treatments and New Approaches
Beyond vaccines and small-molecule drugs, pipelines are expanding into biologics and transmission-blocking technologies.
Monoclonal antibodies for malaria, such as L9LS, designed by scientists at the NIH, and CIS43LS, developed and manufactured by the Vaccine Research Center (VRC), part of the NIH’s National Institute of Allergy and Infectious Diseases (NIAID), offer a promising, long-acting (three to six months) preventive tool by targeting the Plasmodium falciparum parasite’s sporozoite stage before it causes liver infection.
Recent trial data from Mali shows that L9LS was protective against P. falciparum infection and clinical malaria over a period of six months in children.
Similarly, CIS43LS led to durable protection against P. falciparum infection in a Phase II trial.
Some next-generation approaches are focusing on blocking transmission from humans back to mosquitoes, which could help interrupt the cycle of infection entirely, an important shift from purely treating disease toward eliminating transmission.
Other approaches include biological methods such as the Wolbachia method, which introduces Wolbachia bacteria into Aedes aegypti mosquitoes to reduce the transmission of diseases like malaria, dengue, Zika and chikungunya. The bacteria hinder virus growth inside the mosquito, while also causing reproductive incompatibility, either replacing the wild population or reducing its size.
Despite the exciting advances in the field, experts caution that access, affordability and health system capacity will determine whether they translate into real-world impact, particularly in the regions where malaria burden remains highest.










Join or login to leave a comment
JOIN LOGIN