April marks Irritable Bowel Syndrome (IBS) Awareness Month 2026, a time to raise visibility around one of the most common yet misunderstood gastrointestinal conditions.
Affecting an estimated 10% to 15% of people worldwide, IBS can significantly impact quality of life, yet many patients continue to go undiagnosed or undertreated, and remain unaware that their symptoms have a medical explanation.
In 1997, the International Foundation for Gastrointestinal Disorders (IFFGD) designated April as IBS Awareness Month. During this month, the Foundation says it works to focus attention on important health messages about IBS diagnosis, treatment and quality of life issues.
The IFFGD’s theme of IBS Awareness Month 2026 centers on “raising awareness about the hidden realities of IBS and the profound impact these symptoms have on daily life.”
IBS Awareness Month 2026 highlights the importance of education, reducing stigma and patient empowerment. Because IBS is a “functional” disorder, meaning it doesn’t show up clearly on standard diagnostic tests, patients are sometimes dismissed or told their symptoms are “just stress.”
The periwinkle blue ribbon is often used to symbolize IBS awareness, and World IBS Day is observed on April 19.
This month serves as a reminder that IBS is a complex disorder, and that better awareness can lead to earlier diagnosis, improved care and more compassionate conversations.
What Is IBS?
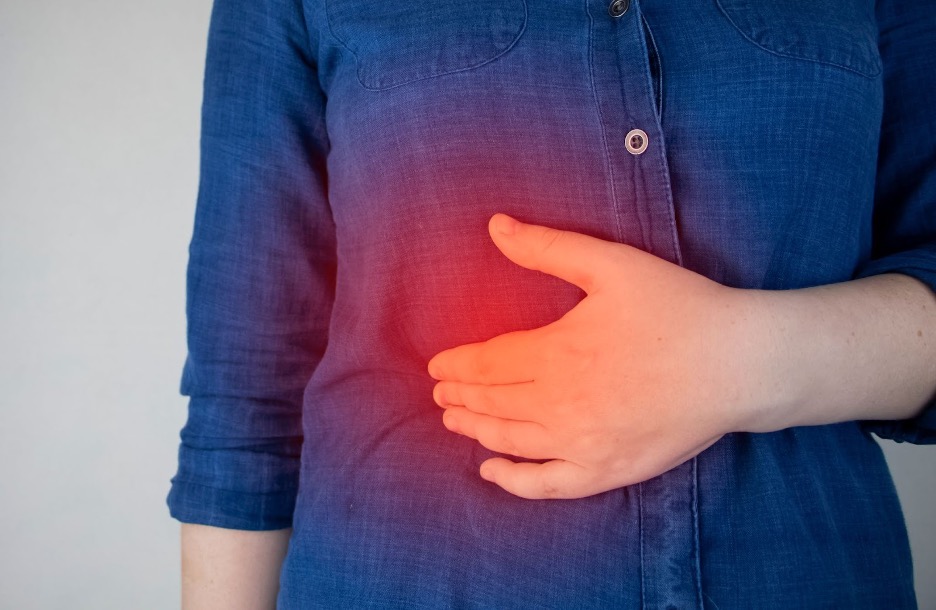
IBS is a chronic condition characterized by abdominal pain, bloating and altered bowel habits (constipation, diarrhea or both).
It’s typically categorized into subtypes: IBS-C (constipation-predominant); IBS-D (diarrhea-predominant); IBS-M (mixed).
While the exact cause isn’t fully understood, research points to a combination of factors, including gut motility issues, visceral hypersensitivity, microbiome imbalances and potential dysregulation of the gut-brain axis.
While no cure exists, management includes:
- Dietary changes: Identifying triggers, often using a low-FODMAP diet, which limits foods like garlic, onions and certain fruits. FODMAP stands for Fermentable Oligo-, Di-, Mono-saccharides And Polyols. It’s a group of short-chain carbohydrates and sugar alcohols that are poorly absorbed in the small intestine. Because they’re not fully digested, they can ferment in the gut, producing gas and drawing water into the intestine. This can lead to bloating, abdominal pain and changes in bowel habits, symptoms common in IBS.
- Lifestyle adjustments: Stress reduction techniques (yoga, meditation) are crucial, as pertaining to the gut-brain connection.
- Medications: Treatments to address specific symptoms like cramping, constipation or diarrhea.
The IBS Treatment Market
The market for IBS therapies remains relatively modest compared to other gastrointestinal disease areas, but it is steadily growing as awareness, diagnosis rates and treatment options expand.
The global IBS treatment market was valued at $3.64 billion in 2024 and is projected to reach $6.02 billion by 2030, growing at a compound annual growth rate (CAGR) of 8.8% from 2025 to 2030.
Despite this, the space is still dominated by a small number of established therapies, none of which have quite exceeded blockbuster-level revenues, underscoring both the fragmented nature of the market and the persistent unmet need among patients.
Among the leading drugs, Linzess (linaclotide), marketed by AbbVie and originally developed by Ironwood Pharmaceuticals, continues to dominate the IBS-C segment. The drug generated approximately $864.5 million in US net sales in 2025, placing it close to blockbuster status.
Another major player is Xifaxan (rifaximin), manufactured and marketed by Salix Pharmaceuticals, a subsidiary of Bausch Health Companies Inc., which is widely used for IBS-D. The drug has historically generated over $800 million annually and remains a key therapy due to its microbiome-targeting mechanism. In addition to IBS-D, the non-absorbable, broad-spectrum oral antibiotic is used to treat traveler’s diarrhea and to reduce the risk of hepatic encephalopathy recurrence, acting directly in the gastrointestinal tract to kill bacteria. The drug’s long-term market position may be challenged by increasing generic competition and pricing pressures.
Newer entrants are also beginning to carve out meaningful market share. Ardelyx’s Ibsrela (tenapanor) is a first-in-class, minimally absorbed, oral medication approved to treat IBS-C in adults. It works by inhibiting the sodium/hydrogen exchanger 3 (NHE3) in the gut, reducing sodium absorption and increasing water secretion, resulting in softer stool and reduced abdominal pain.
Ibsrela was approved for IBS-C in adults in 2019 and as Xphozah in 2023 for reducing serum phosphorus in chronic kidney disease (CKD) patients on dialysis. Its projected US sales were approximately $240 million to $250 million in 2025. Sales of the drug in 2025 totaled $274.2 million, an impressive growth of 73% compared to the $158.3 million reported for the full year 2024.
Meanwhile, Trulance (plecanatide), also marketed by Salix, competes directly with Linzess in IBS-C but holds a smaller share of the market.
In the IBS-D category, Viberzi (eluxadoline) remains an important option, although its growth has been more limited due to safety considerations and prescribing restrictions. Viberzi is designed to reduce abdominal pain and improve stool consistency, with a serious, albeit rare, risk of sphincter of Oddi spasm. Viberzi is a trademark of Furiex Pharmaceuticals, Inc., an Allergan affiliate, and was originally developed by Actavis and is also associated with Patheon Pharmaceuticals and Forest Pharmaceuticals.
The drug faces potential pressure from emerging generic competition. Zydus Lifesciences received FDA approval for its generic version of Viberzi in March 2025, which could introduce significant competition to the market.
Looking ahead, emerging treatments targeting the gut microbiome, gut-brain axis and specific molecular pathways could reshape the competitive IBS treatment landscape, offering both improved patient outcomes and new commercial opportunities in a space that has historically been underserved.














Join or login to leave a comment
JOIN LOGIN