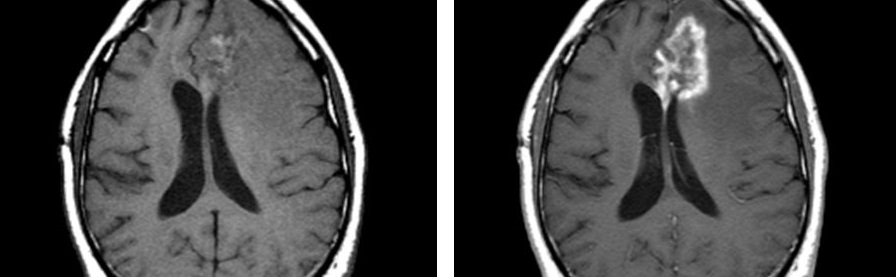
A large majority of the work being done in oncology drug development relates in one way or another to manipulation of the immune system. This approach has many benefits for patients, including the promise of a more robust and long-lasting response to therapy combined with more tolerable side-effect profiles relative to traditional chemotherapy or targeted therapies. However, immune oncology presents unique challenges from an imaging standpoint, prominently including the possibility of pseudo-progression. This talk will review the history of immunotherapy, discuss a range of approaches currently in the clinic or in development, and explore the unique challenges that these new treatment options present in the clinical trials setting.
This webinar is Part 2 of a series on Imaging in Oncology:
Click here for Part 1: Advanced Imaging in Oncology (upcoming on Nov 2/21)
Click here for Part 3: Deep Learning for Image Analysis (upcoming on Nov 8/21)
Speaker

Edward Ashton, PhD, VP, Oncology Imaging, Philips BioTel Research
Ed Ashton serves as the Vice President of Oncology Imaging for Philips BioTel Research. In this role, he has provided technical leadership on more than 200 clinical trials in oncology, neurology, and cardiology over the past twenty years. Dr. Ashton is a frequent speaker at international imaging conferences, and has authored many peer-reviewed publications describing his research. Prior to joining BioTel Research, Dr. Ashton was a lead signal processing engineer at The MITRE Corporation in McLean, VA. Earlier in his career, he spent three years as a research engineer with the Naval Research Laboratory, where he received the Alan Berman Research Publication Award and was nominated for the Edison Award for Applied Science.
Who Should Attend?
This webinar will benefit medical and non-medical professionals in the biopharmaceutical industry, especially those supporting oncology drug development with roles in:
- Clinical Research
- Clinical Development
- Medical Affairs
- Clinical Operations
- Project Management
- Regulatory Affairs
Xtalks Partner
Philips BioTel Research
Philips BioTel Research is an industry leader in medical imaging and cardiovascular safety testing for clinical trials. Philips BioTel Research offers global operational support for cardiovascular monitoring in all therapeutic areas, and advanced imaging services in oncology, cardiovascular, metabolic, musculoskeletal, neurologic, and medical device studies. Their experienced research team comprises key opinion leaders, board-certified cardiologists and radiologists, sub-specialty scientists, and highly trained technicians — who acquire, evaluate, and report high-quality data through an efficient, cloud-based infrastructure. Many biopharmaceutical trial sponsors rely on our scientific leaders to help design and optimize their protocols and statistical analysis plans. We have conducted clinical trials contributing to the market approval of nearly 100 new drugs to date.
You Must Login To Register for this Free Webinar
Already have an account? LOGIN HERE. If you don’t have an account you need to create a free account.
Create Account