MiniMed has secured FDA clearance for its latest insulin delivery system, the MiniMed Flex.
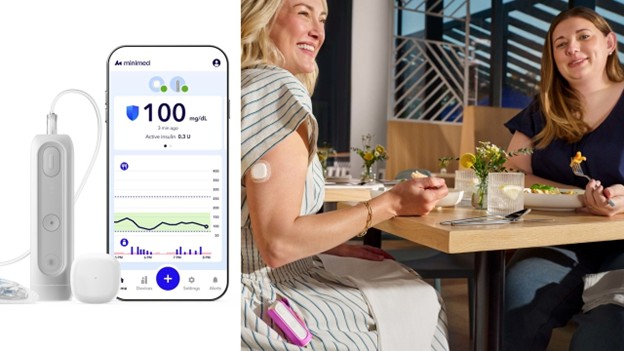
The newly cleared device, described as the company’s smallest insulin pump to date, introduces a fully smartphone-controlled design.
At roughly half the size of MiniMed’s previous model, the 780G pump, the MiniMed Flex is described as being about the size of two stacked insulin vials, small enough to be worn discreetly under clothing.
Unlike traditional insulin pumps, the pocket-sized Flex eliminates the need for an onboard screen. Instead, it is fully controlled through a smartphone app, allowing users to manage insulin delivery, monitor glucose trends and adjust settings from their personal devices.
The MiniMed Flex integrates with continuous glucose monitoring (CGM) and the company’s automated insulin delivery (AID) system, enabling real-time adjustments to insulin dosing.
Like earlier MiniMed systems, the Flex uses algorithm-driven automation to help maintain glucose levels within target range, reducing both hyperglycemia and hypoglycemia without constant manual intervention.
Carrying a 300-unit insulin reservoir, MiniMed Flex is powered by the company’s SmartGuard dosing algorithm with Meal Detection technology, which are also featured in the 780G.
The FDA clearance includes its use in type 1 diabetes for ages seven and older and in type 2 diabetes for adults 18 years of age and older.
In the press release announcing the FDA clearance, MiniMed said the Flex is the company’s “first screenless design created in partnership with people living with diabetes to deliver an intuitive, lifestyle-friendly way to manage the condition.”
MiniMed Flex will support MiniMed’s newest sensor portfolio, which includes the Simplera Sync sensor and the Instinct sensor, made by Abbott.
Medtronic announced in 2025 that it would spin off its roughly $2.8 billion diabetes division into a standalone, publicly traded company to sharpen its focus on higher-margin businesses like cardiovascular and surgical devices, while allowing the diabetes unit to operate more independently and pursue consumer-focused growth.
The MiniMed spinout launched an IPO roadshow at the end of February 2026, raising $560 million by pricing shares at $20, lower than the expected $25 to $28 range. The offering valued the company at roughly $5.61 billion, with Medtronic retaining 90% ownership ahead of a planned full separation.
“At MiniMed, our Mission is to make every day a better day for people with diabetes,” said Que Dallara, Chief Executive Officer of MiniMed.
“MiniMed Flex embodies that promise. It’s designed to work quietly and reliably in the background — advanced automation wrapped in a compact, smartphone‑controlled pump. The result is technology that lets people spend less time managing diabetes and more time living their lives. Securing FDA clearance for our first product as a standalone, public company — just one week after our IPO — marks a major milestone and underscores our commitment to delivering the breakthrough innovation our customers deserve.”
MiniMed’s earlier devices, such as the MiniMed 780G, already introduced hybrid closed-loop functionality. The Flex builds on that foundation by prioritizing usability and form factor, two areas where competitors have been gaining ground.
Earlier this year, MiniMed received FDA clearance for its MiniMed Go smart multiple daily injection (MDI) system, the first and only smart MDI solution that fully integrates personalized insulin dosing with continuous glucose monitoring (CGM) data in a single mobile app.













Join or login to leave a comment
JOIN LOGIN