The Americas’ life sciences sector features fast-growing companies spanning oncology, rare disease, cardiovascular care, radiopharmaceuticals and women’s health.
The Financial Times’ The Americas’ Fastest-Growing Companies 2026 ranking, compiled with Statista, highlights 300 companies across North and South America with the highest compound annual growth rate (CAGR) between 2021 and 2024. To qualify, companies had to be independent, headquartered in the Americas and meet minimum revenue thresholds of $100,000 in 2021 and $1.5 million in 2024.
Within the healthcare and life sciences portion of the list, growth has come from a mix of commercial execution, regulatory approvals, clinical pipeline progress, strategic partnerships and renewed investor interest in platform technologies. The companies below represent some of the fastest-growing pharma, biotech and life sciences businesses in the Americas for 2026 from FT’s list.
Related: Top 20 Most Innovative Biotech Companies of 2026, Per Fast Company
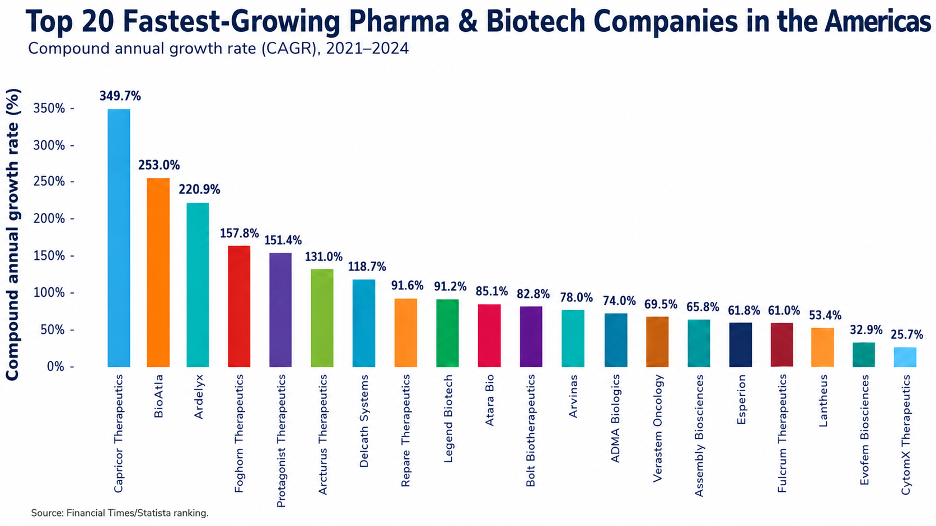
1. Capricor Therapeutics
Location: US
Overall rank: 3
Absolute growth rate (%): 8,994.1
Compound annual growth rate (%): 349.7
Revenue 2024: $22.27 million
Revenue 2021: $244,890
Number of employees 2024: 160
Number of employees 2021: 48
Year founded: 2005
Capricor Therapeutics is a biotechnology company developing cell and exosome-based therapeutics, with a lead focus on deramiocel for Duchenne muscular dystrophy (DMD). In May 2026, Capricor Therapeutics reported that deramiocel’s BLA was under active FDA review, with a PDUFA target action date of August 22, 2026. Capricor also reported that the Phase III HOPE-3 study met its primary endpoint and all Type I error-controlled secondary endpoints. Capricor continues to be engaged in launch preparations, manufacturing expansion and legal action involving its US distribution agreement with Nippon Shinyaku and NS Pharma. Capricor Therapeutics reported no revenue in 2025, compared with approximately $22.27 million in 2024. The company said its prior revenue came from recognition of upfront and milestone payments under its Nippon Shinyaku agreement, which had been fully recognized by the end of 2024.
2. BioAtla
Location: US
Overall rank: 10
Absolute growth rate (%): 4,300
Compound annual growth rate (%): 253
Revenue 2024: $11 million
Revenue 2021: $250,000
Number of employees 2024: 61
Number of employees 2021: 56
Year founded: 2007
BioAtla is an oncology biotech developing conditionally active biologics, including ADCs and immune-oncology candidates. Its pipeline includes ozuriftamab vedotin, a CAB-ROR2 antibody-drug conjugate (ADC) in Phase III development for oropharyngeal squamous cell carcinoma, and mecbotamab vedotin, a CAB-AXL ADC being studied in sarcoma and non-small cell lung cancer (NSCLC). In late 2025, BioAtla and GATC Health announced a $40 million special purpose vehicle transaction to advance ozuriftamab vedotin into a registrational trial, while in 2026, the company launched a formal process to evaluate strategic options to monetize assets. BioAtla reported $2 million in collaboration and other revenue in 2025, down from $11 million in 2024. The 2025 amount reflected a milestone payment from Context Therapeutics, while 2024 revenue was tied to the initial upfront payment under that license agreement.
3. Ardelyx
Location: US
Overall rank: 12
Absolute growth rate (%): 3,204.1
Compound annual growth rate (%): 220.9
Revenue 2024: $333.62 million
Revenue 2021: $10.10 million
Number of employees 2024: 437
Number of employees 2021: 86
Year founded: 2007
Ardelyx is a commercial-stage biopharmaceutical company focused on gastrointestinal and cardiorenal diseases. Its marketed products include Ibsrela (tenapanor) for irritable bowel syndrome (IBS) with constipation and Xphozah (tenapanor) for phosphate reduction in adults with chronic kidney disease (CKD) on dialysis. Ardelyx reported 2025 total revenue of $407.3 million, up from $333.6 million in 2024. Ibsrela revenue grew 73% to $274.2 million, while Xphozah revenue was $103.6 million, down from $160.9 million in 2024.
4. Foghorn Therapeutics
Location: US
Overall rank: 23
Absolute growth rate (%): 1613.6
Compound annual growth rate (%): 157.8
Revenue 2024: $22.60 million
Revenue 2021: $1.32 million
Number of employees 2024: 112
Number of employees 2021: 119
Year founded: 2015
Foghorn Therapeutics is a biotech developing medicines that target abnormal gene expression through its Gene Traffic Control platform. The company’s work centers on chromatin biology and genetically defined cancers. Foghorn has a strategic collaboration with Eli Lilly that includes a 50/50 US co-development and co-commercialization arrangement for a selective SMARCA2 oncology program, including its first-in-class oral SMARCA2 inhibitor FHD-909. In 2026, Foghorn said it and Lilly anticipated evaluating FHD-909 in combination studies in the frontline NSCLC setting. The company chose to continue to focus on its proprietary pipeline and Lilly-partnered programs after discontinuing independent development of FHD-286 in AML. Foghorn reported 2025 collaboration revenue of $30.9 million, up from $22.6 million in 2024. The increase was driven by continued advancement of programs under its Lilly collaboration.
5. Protagonist Therapeutics
Location: US
Overall rank: 25
Absolute growth rate (%): 1,488
Compound annual growth rate (%): 151.4
Revenue 2024: $434.43 million
Revenue 2021: $27.36 million
Number of employees 2024: 98
Number of employees 2021: 92
Year founded: 2006
Protagonist Therapeutics is a biopharmaceutical company developing peptide-based therapies, with major programs in rare hematology and immunology. Its most advanced asset, rusfertide, is a first-in-class investigational hepcidin mimetic being developed with Takeda for polycythemia vera. In March 2026, the FDA accepted the rusfertide NDA and granted Priority Review, with a PDUFA goal date in August 2026. In April 2026, Protagonist exercised its US opt-out right under the Takeda collaboration, giving Takeda exclusive US development and commercialization rights while Protagonist opted for milestone and royalty economics. Protagonist is also partnered with Johnson & Johnson on icotrokinra (brand name Icotyde), which was first approved by the FDA in March 2026 for the treatment of moderate-to-severe plaque psoriasis in adults and adolescents aged 12 and older, making it the first oral IL-23 receptor antagonist in the indication. Protagonist Therapeutics reported 2025 license and collaboration revenue of $46 million, down significantly from $434.4 million in 2024. The decrease reflected the nature of collaboration revenue, as 2024 included major Takeda and J&J-related payments and milestones.
6. Arcturus Therapeutics
Location: US
Overall rank: 34
Absolute growth rate (%): 1,132.4
Compound annual growth rate (%): 131
Revenue 2024: $152.31 million
Revenue 2021: $12.36 million
Number of employees 2024: 176
Number of employees 2021: 177
Year founded: 2013
Arcturus Therapeutics is an mRNA medicines and vaccines company with proprietary RNA and mRNA technologies, including the LUNAR lipid-mediated delivery and STARR self-amplifying mRNA platforms. Its COVID-19 vaccine Kostaive became the first approved self-amplifying mRNA COVID-19 vaccine, with approvals including Japan and the EU through its partnership with CSL. Arcturus and CSL are also working on self-amplifying mRNA vaccines for influenza and other infectious disease targets. The FDA granted Fast Track designation in 2025 to Arcturus’ H5N1 pandemic influenza vaccine candidate ARCT-2304. Arcturus reported 2025 revenue of $82 million, down by $70.3 million from 2024. The company’s revenue primarily came from license fees, consulting and technology transfer fees, reservation fees and collaborative payments from biopharma partners.
7. Delcath Systems
Location: US
Overall rank: 38
Absolute growth rate (%): 946.6
Compound annual growth rate (%): 118.7
Revenue 2024: $37.21 million
Revenue 2021: $3.56 million
Number of employees 2024: 96
Number of employees 2021: 55
Year founded: 1987
Delcath Systems is an interventional oncology company focused on liver-directed therapies for cancers that metastasize to the liver. Its lead commercial product, Hepzato Kit (melphalan for injection/hepatic delivery system), received FDA approval in 2023 as a liver-directed treatment for adult patients with metastatic uveal melanoma with unresectable hepatic metastases. In 2026, Delcath highlighted the inclusion of Chemosat hepatic delivery system and Hepzato Kit-related approaches in guideline discussions, while Hepzato continued to drive commercial growth in the US. Delcath Systems’ 2025 earnings totaled $85.2 million compared to $37.21 million in 2024. Revenue in 2025 included $78.8 million in sales of Hepzato in the US and $6.4 million of Chemostat in Europe.
8. Repare Therapeutics
Location: Canada
Overall rank: 52
Absolute growth rate (%): 603.6
Compound annual growth rate (%): 91.6
Revenue 2024: $53.48 million
Revenue 2021: $7.60 million
Number of employees 2024: 129
Number of employees 2021: 152
Year founded: 2016
Repare Therapeutics is a precision oncology company based in Montreal, Canada, that focuses on synthetic lethality and DNA damage repair targets. Its pipeline has included clinical-stage programs such as lunresertib, camonsertib and other targeted oncology candidates. In 2025, Repare Therapeutics entered a definitive agreement to be acquired by XenoTherapeutics, with shareholders expected to receive an estimated $1.82 per share in cash plus one contingent value right (CVR) tied to future proceeds from partnerships and asset monetization. The transaction is expected to close in the first quarter of 2026, after which Repare would become privately held and delist from Nasdaq. Also in 2025, Repare Therapeutics entered an exclusive worldwide licensing agreement with Debiopharm for lunresertib, a PKMYT1 inhibitor, enabling continued development of the program in difficult-to-treat solid tumors. The company has also out-licensed discovery programs and continued to refine its pipeline after regaining global rights to camonsertib, the company’s oral small molecule ATR (Ataxia-Telangiectasia and Rad3-related protein kinase) inhibitor, from Roche in 2024. Repare Therapeutics reported $11.6 million in revenue for Q3 2025, while revenue from collaboration agreements was $300,000 for the first six months of 2025, down from $53.5 million in the first half of 2024.
9. Legend Biotech
Location: US
Overall rank: 53
Absolute growth rate (%): 598.6
Compound annual growth rate (%): 91.2
Revenue 2024: $627.24 million
Revenue 2021: $89.79 million
Number of employees 2024: 2,500
Number of employees 2021: 1,071
Year founded: 2014
Legend Biotech is a cell therapy company best known for Carvykti (ciltacabtagene autoleucel), a BCMA-targeted CAR-T therapy for multiple myeloma developed with Johnson & Johnson. Carvykti was first FDA-approved in 2022 for adults with relapsed or refractory multiple myeloma after four prior lines of therapy, and in 2024, it received FDA approval for earlier-line use after at least one prior line of therapy in certain patients. The product has become a major growth driver, with Legend Biotech reporting preliminary first-quarter 2026 Carvykti net trade sales of about $597 million based on Janssen data. Legend Biotech reported 2025 total revenue of $1.03 billion, up from $627.3 million in 2024. Collaboration revenue rose to $944.8 million, driven by increased revenue from Carvykti sales under its Janssen collaboration. Carvykti net trade sales were approximately $1.9 billion for the full year of 2025.
10. Atara Bio
Location: US
Overall rank: 59
Absolute growth rate (%): 533.9
Compound annual growth rate (%): 85.1
Revenue 2024: $128.94 million
Revenue 2021: $20.34 million
Number of employees 2024: 153
Number of employees 2021: 578
Year founded: 2012
Atara Bio is a T-cell immunotherapy company developing allogeneic Epstein-Barr virus T-cell therapies for cancer and autoimmune disease. Its lead asset, tabelecleucel or tab-cel, is marketed as Ebvallo in Europe for EBV-positive post-transplant lymphoproliferative disease after prior therapy. In the US, however, the program has faced regulatory setbacks. In January 2026, the FDA declined to approve the therapy, citing issues with the Phase III ALLELE study’s ability to support approval. Atara Bio and partner Pierre Fabre continued regulatory engagement through a Type A meeting, which proved to be fruitful as the FDA has now agreed that the companies may resubmit Ebvallo using the ALLELE single-arm study with an appropriate historical control, reversing its earlier position that the trial design could not support approval. The partners plan to submit updated data and provide another regulatory update in Q3 2026. Atara reported 2025 commercialization revenue of $120.8 million, compared with $128.9 million in 2024. Atara Bio also reported net income of $32.7 million for fiscal 2025, compared with a net loss of $85.4 million in 2024.
11. Bolt Biotherapeutics
Location: US
Overall rank: 62
Absolute growth rate (%): 510.3
Compound annual growth rate (%): 82.8
Revenue 2024: $7.69 million
Revenue 2021: $1.26 million
Number of employees 2024: 52
Number of employees 2021: 91
Year founded: 2015
Bolt Biotherapeutics is an oncology biotech developing immune-stimulating antibody conjugates through its Boltbody ISAC platform. The company’s approach is designed to combine tumor targeting with innate immune activation. In 2026, Bolt said its collaborations with Genmab and Toray remain centered on its Boltbody platform, while the rest of its pipeline was placed on hold, including BDC-3042, a Dectin-2 agonist that completed a first-in-human Phase I dose-escalation trial. The company is also expecting initial clinical data in Q3 2026 for its immune-stimulating antibody conjugate BDC-4182 (targeting claudin 18.2) from an ongoing Phase I/II study after a strong immune response was observed at the initial dose levels. This led the company to modify the clinical trial protocol to allow for step-up dosing, which has been successfully used commercially for T-cell engagers, the company explained. BDC-4182 preclinical data support this approach. Bolt Biotherapeutics reported 2025 collaboration revenue of $7.7 million, essentially flat with 2024. The company said 2025 revenue reflected continued progress in collaborations as it fulfilled performance obligations to partners.
12. Arvinas
Location: US
Overall rank: 69
Absolute growth rate (%): 464
Compound annual growth rate (%): 78
Revenue 2024: $263.4 million
Revenue 2021: $46.7 million
Number of employees 2024: 430
Number of employees 2021: 280
Year founded: 2013
Arvinas is a biotech company known for its targeted protein degradation platform. In May 2026, Arvinas and Pfizer received FDA approval for Veppanu (vepdegestrant) for adults with ER-positive, HER2-negative, ESR1-mutated advanced or metastatic breast cancer following at least one line of endocrine therapy. Veppanu became the first FDA-approved PROTAC (proteolysis targeting chimeratherapy). Shortly after the approval, Rigel Pharmaceuticals announced a global licensing agreement for the drug. Arvinas reported full-year 2025 revenue of $262.6 million, nearly flat from $263.4 million in 2024, while narrowing its net loss to $80.8 million from $198.9 million the year prior. Arvinas ended 2025 with $685.4 million in cash, cash equivalents and marketable securities, which it said should fund operations into the second half of 2028.
13. ADMA Biologics
Location: US
Overall rank: 74
Absolute growth rate (%): 426.9
Compound annual growth rate (%): 74
Revenue 2024: $426.45 million
Revenue 2021: $80.94 million
Number of employees 2024: 685
Number of employees 2021: 527
Year founded: 2004
ADMA Biologics is a biopharmaceutical company focused on plasma-derived immunoglobulin products for immunodeficient patients. Its portfolio includes Asceniv and Bivigam, both 10% liquid intravenous immune globulin products used in primary humoral immunodeficiency. ADMA Biologics has benefited from strong demand for ASCENIV, reporting first-quarter 2026 revenue of $114.5 million, with ASCENIV revenue up 28% year-over-year. ADMA Biologics reported record financial results for the full year 2025 with $510 million in total revenue (a 20% year-over-year increase) driven by strong Asceniv sales. The company has also invested in manufacturing and yield improvements, including a previously FDA-approved production process designed to increase immunoglobulin output from the same plasma volume. ADMA Biologics has been in some hot water recently, with an investigation into potential securities claims. The company is facing allegations that it may have issued materially misleading business information. A recent legal notice cites a report from short seller Culper Research alleging “channel stuffing” to inflate revenue growth, after which ADMA’s stock reportedly fell 16.6%.
14. Verastem Oncology
Location: US
Overall rank: 79
Absolute growth rate (%): 387.1
Compound annual growth rate (%): 69.5
Revenue 2024: $10 million
Revenue 2021: $2.05 million
Number of employees 2024: 78
Number of employees 2021: 48
Year founded: 2010
Verastem Oncology is a biopharmaceutical company developing and commercializing therapies for RAS/MAPK pathway-driven cancers. In 2025, the FDA granted accelerated approval to Verastem’s Avmapki Fakzynja Co-pack, a combination of avutometinib and defactinib, for adults with KRAS-mutated recurrent low-grade serous ovarian cancer who have received prior systemic therapy. The approval represented the first FDA-approved treatment specifically for this rare ovarian cancer subtype. In 2026, Verastem Oncology reported two-year follow-up data from the Phase II RAMP 201 study, showing sustained clinical benefit with the combination. Verastem reported $30.9 million in total revenue for the full year 2025, generated from the launch of the Avmapki Fakzynja Co-pack. The 2025 period saw no product revenue in the first few months prior to the May launch. While Verastem did not record product revenue in 2024, it generated revenue from milestone payments related to oncology drug Copiktra (duvelisib) following the 2020 sale of the asset to Secura Bio. Similarly, the company’s 2021 revenue came from milestones, royalties and transition services tied to the Copiktra sale.
15. assemblybio
Location: US
Overall rank: 84
Absolute growth rate (%): 356
Compound annual growth rate (%): 65.8
Revenue 2024: $28.52 million
Revenue 2021: $6.25 million
Number of employees 2024: 73
Number of employees 2021: 70
Year founded: 2005
Assembly Biosciences is a virology-focused biotech company developing small-molecule therapeutics for viral diseases, including herpesvirus, hepatitis B virus and hepatitis delta virus infections. The company has a major collaboration with Gilead Sciences focused on novel antiviral therapies, with several programs, including a helicase-primase inhibitor program for recurrent genital herpes, licensed to Gilead for further development and commercialization. In 2026, Assembly highlighted positive Phase Ib data for its long-acting helicase primase inhibitor candidates ABI-5366 and ABI-1179, while Gilead returned rights to one HBV antiviral program but continued its broader partnership with Assembly on other assets. In 2025, Assembly Bio’s revenue rose to $72.3 million from $28.5 million in 2024, driven by Gilead collaboration funding, as the company currently has no approved commercial products.
16. Esperion
Location: US
Overall rank: 88
Absolute growth rate (%): 323.6
Compound annual growth rate (%): 61.8
Revenue 2024: $332.31 million
Revenue 2021: $78.45 million
Number of employees 2024: 304
Number of employees 2021: 218
Year founded: 2008
Esperion Therapeutics is a cardiovascular-focused pharmaceutical company best known for its oral non-statin LDL cholesterol-lowering products Nexletol (bempedoic acid) and Nexlizet (bempedoic acid and ezetimibe). The company received expanded FDA labels in 2024 for cardiovascular risk reduction and broader LDL-C lowering, helping expand the potential patient population for its bempedoic acid franchise. And in May 2026, Esperion Therapeutics announced it would be acquired by investment firm Archimed in a deal valued at up to $1.1 billion. Esperion Therapeutics reported full-year 2025 total revenue of $403.1 million, up 21% from $332.3 million in 2024.
17. Fulcrum Therapeutics
Location: US
Overall rank: 89
Absolute growth rate (%): 317.5
Compound annual growth rate (%): 61
Revenue 2024: $80 million
Revenue 2021: $19.16 million
Number of employees 2024: 45
Number of employees 2021: 104
Year founded: 2015
Fulcrum Therapeutics is a biopharmaceutical company focused on small molecules for rare diseases, particularly rare hematologic disorders. After discontinuing losmapimod development for facioscapulohumeral muscular dystrophy, Fulcrum has focused on pociredir, an oral fetal hemoglobin (HbF) inducer being studied in sickle cell disease. The drug is a small molecule inhibitor of embryonic ectoderm development (EED) that is designed to downregulate key fetal globin repressors to cause an increase in HbF. In February 2026, Fulcrum reported positive 12-week Phase Ib data for pociredir showing robust fetal hemoglobin induction. In April 2026, the company said it planned to initiate a potential registration-enabling trial in the second half of 2026. Fulcrum reported no revenue in 2025, down from prior collaboration-driven revenue. The company ended 2025 with $352.3 million in cash, cash equivalents and marketable securities, up from $241 million at the end of 2024, largely due to proceeds from a December 2025 public offering.
18. Lantheus
Location: US
Overall rank: 103
Absolute growth rate (%): 260.7
Compound annual growth rate (%): 53.4
Revenue 2024: $1.53 billion
Revenue 2021: $425.21 million
Number of employees 2024: 808
Number of employees 2021: 602
Year founded: 1956
Lantheus is a radiopharmaceutical-focused company with products spanning oncology, cardiology and neurology. It is best known for diagnostic imaging agents such as Pylarify, a PSMA-targeted PET imaging agent for prostate cancer, and Definity, an ultrasound-enhancing agent. In March 2026, the FDA approved Pylarify TruVu, a new formulation of piflufolastat F 18 injection expected to support higher radioactive concentrations and more efficient distribution. Commercial availability of the agent is planned for the fourth quarter of 2026. In April 2025, Lantheus completed its acquisition of Evergreen Theragnostics and of Life Molecular Imaging in July 2025, bolstering its pipeline with assets like Neuraceq (florbetaben F18 injection) from Life, a globally approved F-18 PET imaging agent used to detect beta-amyloid plaques in Alzheimer’s disease patients. Lantheus reported 2025 worldwide revenue of $1.54 billion compared with $1.53 billion in 2024. Pylarify generated $989.1 million in 2025, down 6.5% from 2024.
19. Evofem Biosciences
Location: US
Overall rank: 161
Absolute growth rate (%): 134.9
Compound annual growth rate (%): 32.9
Revenue 2024: $19.36 million
Revenue 2021: $8.24 million
Number of employees 2024: 32
Number of employees 2021: 119
Year founded: 2007
Evofem Biosciences is a women’s health biopharmaceutical company commercializing sexual and reproductive health products. Its lead product, Phexxi, is an FDA-approved, non-hormonal, “on-demand” prescription contraceptive vaginal gel that works to prevent pregnancy by maintaining normal vaginal pH (3.5-4.5), which reduces sperm motility. Evofem also acquired US commercial rights to Solosec from Lupin in 2024, expanding its portfolio into bacterial vaginosis and trichomoniasis. In 2026, Evofem announced an exclusive agreement for the distribution and commercialization of Solosec in Sub-Saharan Africa. Evofem reported 2025 net sales of $20.2 million, up 4% from $19.4 million in 2024, marking its fifth consecutive year of net sales growth. The company said growth was supported by Phexxi and a full year of Solosec sales following its 2024 acquisition and commercial relaunch.
20. CytomX Therapeutics
Location: US
Overall rank: 185
Absolute growth rate (%): 98.5
Compound annual growth rate (%): 25.7
Revenue 2024: $138.10 million
Revenue 2021: $69.57 million
Number of employees 2024: 119
Number of employees 2021: 174
Year founded: 2008
CytomX Therapeutics is an oncology biotech company developing conditionally activated biologics through its Probody therapeutic platform. Its approach is designed to localize biologic activity to the tumor microenvironment, potentially widening the therapeutic window for targets that may be difficult to drug systemically. In March 2026, CytomX reported positive Phase I expansion data for varsetatug masetecan, an EpCAM-targeted Probody ADC, in late-line metastatic colorectal cancer. CytomX Therapeutics reported 2025 total revenue of $76.2 million, down from $138.1 million in 2024. The decrease was mainly attributed to the completion of performance obligations under its Bristol Myers Squibb collaboration and lower recognized revenue from other collaborations.











Join or login to leave a comment
JOIN LOGIN